The equation of state for a gas is given by $PV = nRT + \alpha V$, where $n$ is the number of moles and $\alpha $ is a positive constant. The initial temperature and pressure of one mole of the gas contained in a cylinder are $T_o$ and $P_o$ respectively. The work done by the gas when its temperature doubles isobarically will be
JEE MAIN 2014, Diffcult
${P_0}{V_0} = nR{T_0}$
${P_0}V = NRT$
${T_f} = 2{T_0}$
$W = \int {PdV} $
$ = \int {\left( {\frac{{nRT}}{V} + \alpha } \right)dV} $
$PV = nRT + \alpha V$
$\int {PdV = \int\limits_{{T_0}}^{2{T_0}} {nRdT + \int\limits_{{V_1}}^{{V_1}} {\alpha dV} } } $
$ = nR{T_0} + \alpha \,{V_i}$
$ = nR{T_0} + \alpha \left( {\frac{{nR{T_0}}}{{{P_0}}}} \right)$
$ = nR{T_0} \left( {1 + \frac{\alpha }{{{P_0}}}} \right)$
$PV = nRT + \alpha V$
$\int {PdV = \int {nRdT + \int {\alpha dV} } } $
$W = nR{T_0} + \alpha \left[ {\frac{{nR{T_0}}}{{{P_0} - \alpha }}} \right]$
$W = nR{T_0}\left[ {1 + \frac{\alpha }{{{P_0} - \alpha }}} \right]$
$ = n{R_0}{T_0}\left[ {\frac{{{P_0}}}{{{P_0} - \alpha }}} \right]$
$ = \frac{{nR{T_0}{P_0}}}{{{P_0} - \alpha }}$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionAn adiabatic process occurs at constant
- 2A Carnot engine absorbs an amount $Q$ of heat from a reservoir at an absolute temperature $T$ and rejects heat to a sink at a temperature of $T/3$ . The amount of heat rejected isView Solution
- 3A sample of $0.1\, g$ of water at $100^o C$ and normal pressure $(1.013 \times 10^5 N m^{-2} )$ requires $54\ cal $ of heat energy to convert to steam at $100^o C.$ If the volume of the steam produced is $167.1 \,cc,$ the change in internal energy of the sample, is ....... $J$View Solution
- 4In an Isothermal change, the change in pressure and volume of a gas can be represented for three different temperature; $T _3 > T _2 > T _1$ as :View Solution
- 5One mole of an ideal gas at an initial temperature of $T\, K$ does $6 R$ joules of work adiabatically. If the ratio of specific heats of this gas at constant pressure and at constant volume is $5/3$, the final temperature of gas will beView Solution
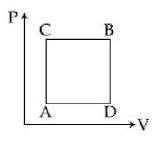
- 6A gas can be taken from $A$ to $B$ via two different processes $ACB$ and $ADB$. When path $ACB$ is used $60\, J$ of heat flows into the system and $30\, J$ of work is done by the system. If path $ADB$ is used work down by the system is $10\, J$. the heat flow into the system in path $ADB$ is ..... $J$View Solution
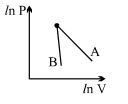
- 7The figure, shows the graph of logarithmic reading of pressure and volume for two ideal gases $A$ and $B$ undergoing adiabatic process. From figure it can be concluded thatView Solution
- 8How much work to be done in decreasing the volume of and ideal gas by an amount of $2.4 \times {10^{ - 4}}{m^3}$ at normal temperature and constant normal pressure of .......$joule$ $1 \times {10^5}N/{m^2}$View Solution
- 9A Carnot engine absorbs $1000\,J$ of heat energy from a reservoir at $127\,^oC$ and rejects $600\,J$ of heat energy during each cycle. The efficiency of engine and temperature of sink will beView Solution
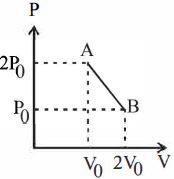
- 10'$n$' moles of an ideal gas undergoes a process $A \rightarrow B$ as shown in the figure. The maximum temperature of the gas during the process will beView Solution