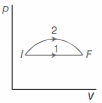
An ideal gas undergoes change in its state from the initial state $I$ to the final state $F$ via two possible paths as shown below. Then,

KVPY 2018, Advanced
$(a,c)$ Given paths over $p-V$ graph are
As in both processes, initial and final points are same, change in internal cnergy (which is not a path function) is same. So, option $(a)$ is correct.
- Both processes are expansion process. So, heat is absorbed in both $1$ and $2$ . Hence, option $(b)$ is incorrect.
- Now, consider isotherms over $p-V$ graph given, we clearly see that for path $2$ temperature increases and then decreases. So, option $(c)$ is correct.
- Area enclosed by path $2$ in $p-V$ graph is larger.
So, work done is more in path $2$. Hence, option $(d)$ is incorrect.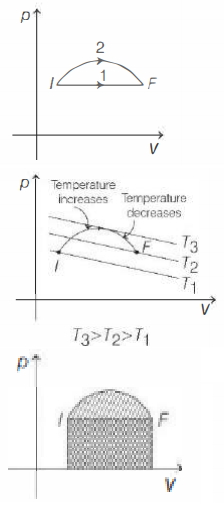
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1The volume of an ideal gas is $1$ litre and its pressure is equal to $72cm$ of mercury column. The volume of gas is made $900\, cm^3$ by compressing it isothermally. The stress of the gas will be ...... $cm$ (mercury)View Solution
- 2The initial pressure and volume of an ideal gas are $P_0$ and $V_0$. The final pressure of the gas when the gas is suddenly compressed to volume $\frac{ V _0}{4}$ will be (Given $\gamma=$ ratio of specific heats at constant pressure and at constant volume)View Solution
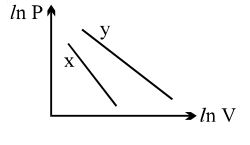
- 3For two different gases $X$ and $Y$, having degrees of freedom $f_1$ and $f_2$ and molar heat capacities at constant volume $C_{V1}$ and $C_{V2}$ respectively, the ln $P$ versus ln $V$ graph is plotted for adiabatic process, as shownView Solution
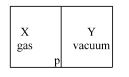
- 4A closed container is fully insulated from outside. One half of it is filled with an ideal gas $X$ separated by a plate Pfrom the other half $Y$ which contains a vacuum as shown in figure. When $P$ is removed, $X$ moves into $Y$. Which of the following statements is correct?View Solution
- 5View SolutionIn which of the following processes, heat is neither absorbed nor released by a system ?
- 6If minimum possible work is done by a refrigerator in converting $100\; grams$ of water at $0^{\circ} C$ to ice, how much heat (in calories) is released to the surrounding at temperature $27^{\circ} C$ (Latent heat of ice $=80 Cal / gram$ ) to the nearest integer?View Solution
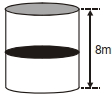
- 7A thermally isolated cylindrical closed vessel of height $8 m$ is kept vertically. It is divided into two equal parts by a diathermic $($perfect thermal conductor$)$ frictionless partition of mass $8.3 kg$. Thus the partition is held initially at a distance of $4 m$ from the top, as shown in the schematic figure below. Each of the two parts of the vessel contains $0.1$ mole of an ideal gas at temperature $300 K$. The partition is now released and moves without any gas leaking from one part of the vessel to the other. When equilibrium is reached, the distance of the partition from the top $($in $m )$ will be $. . . . . ($take the acceleration due to gravity $=10 ms ^{-2}$ and the universal gas constant $=8.3 J mol ^{-1} K ^{-1} ).$View Solution
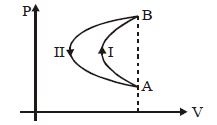
- 8In a cyclic process, a gas is taken from state $A$ to $B$ via path $-I$ as shown in the indicator diagram and taken back to state $A$ from state $B$ via path $-II$ . In the complete cycleView Solution
- 9An ideal heat engine exhausting heat at $77\,^oC$. To have a $30\%$ efficiency. It must take heat at...... $^oC$View Solution
- 10When a system is taken from state $i$ to state $f$ along the path $iaf$, it is found that $Q=50$ $cal$ and $W=20$ $cal$ Along the path $ibf\ Q = 36\ cal. \ W$ along the path $ibf$ is ....... $ cal$View Solution