A Carnot engine with sink's temperature at $17\,^oC$ has $50\%$ efficiency. By how much should its source temperature be changed to increases its efficiency to $60\%$ ?...... $K$
Medium
$\eta=1-\frac{\mathrm{T}_{2}}{\mathrm{T}_{1}}$
$\Rightarrow \frac{50}{100}=1-\frac{290}{\mathrm{T}_{1}} \Rightarrow \mathrm{T}_{1}=580 \mathrm{K}$
$\Rightarrow \frac{60}{100}=1-\frac{290}{\mathrm{T}_{1}^{\prime}} \Rightarrow \mathrm{T}_{1}^{\prime}=725 \mathrm{K}$
$\Rightarrow \Delta \mathrm{T}=\mathrm{T}_{1}^{\prime}-\mathrm{T}_{1}=145 \mathrm{K}$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A container of volume $1{m^3}$is divided into two equal compartments by a partition. One of these compartments contains an ideal gas at $300 K$. The other compartment is vacuum. The whole system is thermally isolated from its surroundings. The partition is removed and the gas expands to occupy the whole volume of the container. Its temperature now would be ..... $K$View Solution
- 2Consider two containers $A$ and $B$ containing identical gases at the same pressure, volume and temperature. The gas in container $A$ is compressed to half of its original volume isothermally while the gas in container $B$ is compressed to half of its original value adiabatically. The ratio of final pressure of gas in $B$ to that of gas in $A$ isView Solution
- 3The figure shows a process $AB$ undergone by $2$ moles of an ideal diatomic gas. The process $AB$ is in such a way that $VT =$ constant. $T_1 = 300 K $and $T_2 = 500 K$ ( $R = $ gas constant)View Solution
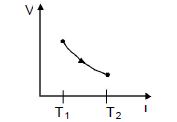
- 4View SolutionAn adiabatic process occurs at constant
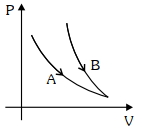
- 5Choose the correct statement for processes $A$ & $B$ shown in figure.View Solution
- 6In an Isobaric process, the work done by a di$-$atomic gas is $10\, J ,$ the heat given to the gas will be (in $J$)View Solution
- 7View SolutionThe work done in which of the following processes is zero
- 8View SolutionFirst law of thermodynamics is a special case of
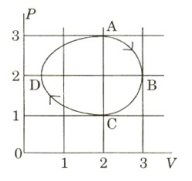
- 9The figure shows the $P-V$ plot of an ideal gas taken through a cycle $ABCDA$. The part $ABC$ is a semi-circle and $CDA$ is half of an ellipse. Then,View Solution
$(A)$ the process during the path $\mathrm{A} \rightarrow \mathrm{B}$ is isothermal
$(B)$ heat flows out of the gas during the path $\mathrm{B} \rightarrow \mathrm{C} \rightarrow \mathrm{D}$
$(C)$ work done during the path $\mathrm{A} \rightarrow \mathrm{B} \rightarrow \mathrm{C}$ is zero
$(D)$ positive work is done by the gas in the cycle $ABCDA$
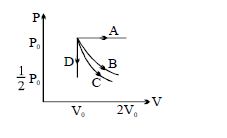
- 10The $PV$ diagram shows four different possible reversible processes performed on a monatomic ideal gas. Process $A$ is isobaric (constant pressure). Process $B$ is isothermal (constant temperature). Process $C$ is adiabatic. Process $D$ is isochoric (constant volume). For which process$(es)$ does the temperature of the gas decrease?View Solution