A Carnot engine whose sink is at $300\, K$ has an efficiency of $40\%.$ By how much should the temperature of source be increased so as to increase its efficiency by $50\%$ of original efficiency ..... $K$
AIPMT 2006, Diffcult
Efficiency of a Carnot engine,$\eta = 1\frac{{{T_2}}}{{{T_1}}}$
$or,\,\,\frac{{{T_2}}}{{{T_1}}} = 1 - \eta = 1 - \frac{{40}}{{100}} = \frac{3}{5}$
$\therefore {T_1} = \frac{5}{3} \times {T_2} = \frac{5}{3} \times 300 = 500\,K.$
$Increases\,in\,efficiency = 50\% \,of\,40\% = 20\% $
$New\,efficiency,\eta ' = 40\% + 20\% = 60\% $
$\therefore \,\,\frac{{{T_2}}}{{{T_1}'}} = 1 - \frac{{60}}{{100}} = \frac{2}{5}$
${T_1}' = \frac{5}{2} \times {T_2} = \frac{5}{2} \times 300 = 750\,K.$
$Increases\,in\,temperature\,of\,source = {T_1}' - {T_1}$
$ = 750 - 500 = 250\,K.$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1An ideal gas heat engine operates in Carnot cycle between $227°C$ and $127°C.$ It absorbs $6 \times {10^4}$ cals of heat at higher temperature. Amount of heat converted to work is .........$ \times {10^4}\; cal$View Solution
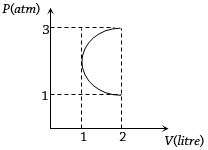
- 2In the $P-V$ diagram shown in figure $ABC$ is a semicircle. The work done in the process $ABC$ isView Solution
- 3An iron rod of heat capacity $C$ is heated to temperature $8T_0$ . It is then put in a cylindrical vessel of adiabatic walls having two moles of air which can be treated as diatomic ideal gas at temperature $T_0$ and closed by a movable piston which is also adiabatic. The atmospheric pressure is $P_0$ . The cylinder with the piston combined have heat capacity $2C$ . Find the equilibrium temperature . (Assume temperature of air to be uniform and equal to vessel at all times) .View Solution
- 4A mass of diatomic gas $(\gamma = 1 .4)$ at a pressure of $2$ atmospheres is compressed adiabatically so that its temperature rises from $27^o C$ to $927^o C.$ The pressure of the gas in the final state is ...... $atm$View Solution
- 5The work of $146\,kJ$ is performed in order to compress one kilo mole of a gas adiabatically and in this process the temperature of the gas increases by $7\,^oC$ . The gas is $(R = 8.3\, J\, mol^{-1}\, K^{-1})$View Solution
- 6The latent heat of vaporization of water is $2240 \,J/gm$. If the work done in the process of vaporization of $1\, gm$ is $168\, J$, then increase in internal energy is .... $J$View Solution
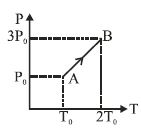
- 7Two moles of helium gas are taken over the cycle $ABCDA$, as shown in the $P-T$ diagram. The net work done on the gas in the cycle $ABCDA$ is ...... $R$View Solution

- 8A Carnot engine working between $300\,K$ and $600\,K$ has work output of $800\, J$ per cycle. What is amount of heat energy supplied to the engine from source per cycle...... $J/cycle$View Solution
- 9Pressure versus temperature graph of an ideal gas is as shown in figure. Density of the gas at point $A$ is ${\rho _0}$. Density at point $B$ will beView Solution
- 10A closed vessel contains $0.1$ mole of a monoatomic ideal gas at $200\, K$. If $0.05$ mole of the same gas at $400\, K$ is added to it, the final equilibrium temperature (in $K$ ) of the gas in the vessel will be closed toView Solution
