At ${27^o}C$ a gas is suddenly compressed such that its pressure becomes $\frac{1}{8}th$ of original pressure. Temperature of the gas will be $(\gamma = 5/3)$
Medium
(d) ${T^\gamma }{P^{1 - \gamma }} = $constant ==> $T \propto {P^{\frac{{\gamma - 1}}{\gamma }}}$
==> $\frac{{{T_2}}}{{{T_1}}} = {\left( {\frac{{{P_2}}}{{{P_1}}}} \right)^{\frac{{\gamma - 1}}{\gamma }}} = {\left( {\frac{1}{8}} \right)^{\frac{{5/3 - 1}}{{5/3}}}}$
${T_2} = 300 \times {\left( {\frac{1}{8}} \right)^{0.4}} = 131K = - 142^\circ C$
==> $\frac{{{T_2}}}{{{T_1}}} = {\left( {\frac{{{P_2}}}{{{P_1}}}} \right)^{\frac{{\gamma - 1}}{\gamma }}} = {\left( {\frac{1}{8}} \right)^{\frac{{5/3 - 1}}{{5/3}}}}$
${T_2} = 300 \times {\left( {\frac{1}{8}} \right)^{0.4}} = 131K = - 142^\circ C$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
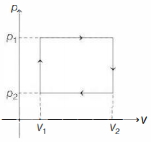
- 1$n$ moles of a van der Waals' gas obeying the equation of state $\left(p+\frac{n^2 a}{V^2}\right)(V-n b)=n R T$, where $a$ and $b$ are gas dependent constants, is made to undergo a cyclic process that is depicted by a rectangle in the $p-V$ diagram as shown below. What is the heat absorbed by the gas in one cycle?View Solution
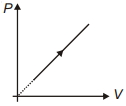
- 2The gas law $\frac{{PV}}{T} = $ constant is true forView Solution
- 3Which of the accompanying $PV$, diagrams best represents an isothermal processView Solution
- 4View SolutionThe work done in which of the following processes is equal to the internal energy of the system?
- 5View SolutionDuring the thermodynamic process shown in figure for an ideal gas
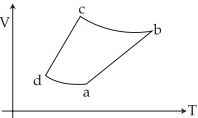
- 6An ideal gas goes through a reversible cycle $a\to b\to c\to d$ has the $V - T$ diagram shown below. Process $d\to a$ and $b\to c$ are adiabatic.... The corresponding $P - V$ diagram for the process is (all figures are schematic and not drawn to scale)View Solution
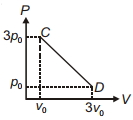
- 7The process $C D$ is shown in the diagram. As system is taken from $C$ to $D$, what happens to the temperature of the system?View Solution
- 8A monoatomic ideal gas, initially at temperature ${T_1},$ is enclosed in a cylinder fitted with a frictionless piston. The gas is allowed to expand adiabatically to a temperature. ${T_2}$ by releasing the piston suddenly. If ${L_1}$ and ${L_2}$ are the lengths of the gas column before and after expansion respectively, then ${T_1}/{T_2}$ is given byView Solution
- 9An ideal gas is taken reversibly around the cycle $a-b-c-d-a$ as shown on the temperature $T$ - entropy $S$ diagram. The most appropriate representation of above cycle on a internal energy $U$ - volume $V$ diagram isView Solution
- 10When the amount of work done is $333\, cal$ and change in internal energy is $167\, cal$, then the heat supplied is ....... $cal$View Solution
