A thermodynamic cycle $xyzx$ is shown on a $V-T$ diagram.
The $P-V$ diagram that best describes this cycle is
(Diagrams are schematic and not to scale)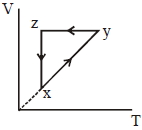
JEE MAIN 2020, Medium
$\mathrm{x} \rightarrow \mathrm{y} \Rightarrow$ Isobaric
$\mathrm{y} \rightarrow \mathrm{z} \Rightarrow$ Isochoric
$\mathrm{z} \rightarrow \mathrm{x} \Rightarrow \mathrm{Isothermal}$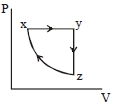
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A polyatomic gas with six degrees of freedom does $25\,\,J$ of work when it is expanded at constant pressure. The heat given to the gas is ..... $J$View Solution
- 2View SolutionThe state of a thermodynamic system is represented by
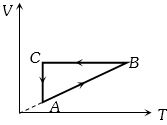
- 3A cyclic process $ABCA$ is shown in the $V-T $ diagram. Process on the $P-V$ diagram isView Solution
- 4Determine efficiency of carnot cycle if in adiabatic expansion volume $3$ times of initial value and $\gamma =1.5$View Solution
- 5View SolutionThe slopes of isothermal and adiabatic curves are related as
- 6An iron rod of heat capacity $C$ is heated to temperature $8T_0$ . It is then put in a cylindrical vessel of adiabatic walls having two moles of air which can be treated as diatomic ideal gas at temperature $T_0$ and closed by a movable piston which is also adiabatic. The atmospheric pressure is $P_0$ . The cylinder with the piston combined have heat capacity $2C$ . Find the equilibrium temperature . (Assume temperature of air to be uniform and equal to vessel at all times) .View Solution
- 7View SolutionAn ideal gas, undergoing adiabatic change, has which of the following pressure temperature relationship?
- 8A gas mixture consists of $8$ moles of argon and $6$ moles of oxygen at temperature $T$. Neglecting all vibrational modes, the total internal energy of the system isView Solution
- 9A gas for which $\gamma = 1.5$ is suddenly compressed to $\frac{1}{4}$ th of the initial volume. Then the ratio of the final to the initial pressure isView Solution
- 10The specific heat of hydrogen gas at constant pressure is ${C_P} = 3.4 \times {10^3}cal/kg{\,^o}C$ and at constant volume is ${C_V} = 2.4 \times {10^3}cal/kg{\,^o}C.$If one kilogram hydrogen gas is heated from ${10^o}C$ to ${20^o}C$ at constant pressure, the external work done on the gas to maintain it at constant pressure isView Solution
