An ideal gas undergoes a polytropic given by equation $P V^n=$ constant. If molar heat capacity of gas during this process is arithmetic mean of its molar heat capacity at constant pressure and constant volume then value of $n$ is ..............
Diffcult
(b)
Polytropic process
$P V^m=$ constant
Given heat capacities is average of $C_P$ and $C_V$ So
$C=\frac{C_P+C_V}{2}$
or $\quad C=\frac{2 C_V+R}{2}$
or $\quad C=\frac{C_V+R}{2} \quad \dots (i)$
Now formula for specific heat of polytropic process is given by
$C=\frac{R}{y-1}+\frac{R}{1-n} \quad \dots (ii)$
or $\frac{R}{y-1}+\frac{R}{2}=\frac{R}{y-1}+\frac{R}{1-n}$ as $C_V=\frac{R}{y-1}$
$\frac{R}{2}=\frac{R}{1-n}$
or $n=-1$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1An ideal gas expands according to the law $P^2 V=$ constant. The internal energy of the gasView Solution
- 2Heat given to a system is $35$ joules and work done by the system is $15$ joules. The change in the internal energy of the system will be ..... $J$View Solution
- 3View SolutionBy opening the door of a refrigerator placed inside a room you
- 4An ideal gas is expanded adiabatically at an initial temperature of $300 K$ so that its volume is doubled. The final temperature of the hydrogen gas is $(\gamma = 1.40)$View Solution
- 5$Assertion :$ The Carnot cycle is useful in understanding the performance of heat engines.View Solution
$Reason :$ The Carnot cycle provides a way of determining the maximum possible efficiency achievable with reservoirs of given temperatures. - 6A cyclic process $ABCD$ is shown in the $p-V$ diagram. Which of the following curves represents the same process if $BC \& DA$ are isothermal processesView Solution
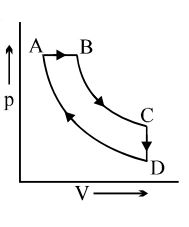
- 7A gas may expand either adiabatically or isothermally. A number of $P-V$ curves are drawn for the two processes over different range of pressure and volume. It will be found thatView Solution
- 8A thermodynamic cycle $xyzx$ is shown on a $V-T$ diagram.View Solution
The $P-V$ diagram that best describes this cycle is
(Diagrams are schematic and not to scale)
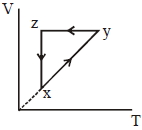
- 9A Carnot's engine working between $400\, K$ and $800\, K$ has a work output of $1200\, J$ per cycle. The amount of heat energy supplied to the engine from the source in each cycle is ........... $J$View Solution
- 10A Carnot engine operating between two reservoirs has efficiency $\frac{1}{3}$. When the temperature of cold reservoir raised by $x$, its efficiency decreases to $\frac{1}{6}$. The value of $x$, if the temperature of hot reservoir is $99^{\circ}\,C$, will be $........\,K$View Solution
