Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1For an isothermal expansion of a perfect gas, the value of $\frac{{\Delta P}}{P}$ is equalView Solution
- 2A sample of gas expands from volume ${V_1}$ to ${V_2}$. The amount of work done by the gas is greatest when the expansion isView Solution
- 3View SolutionPressure versus temperature graph of an ideal gas is shown in figure
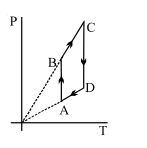
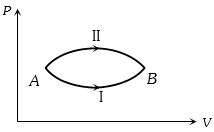
- 4A system goes from $A$ to $B$ via two processes $I$ and $II$ as shown in figure. If $\Delta {U_1}$ and $\Delta {U_2}$ are the changes in internal energies in the processes $I$ and $II$ respectively, thenView Solution
- 5One mole of an ideal gas expands adiabatically from an initial state $\left(T_A, V_0\right)$ to final state $\left(T_f, 5 V_0\right)$. Another mole of the same gas expands isothermally from a different initial state ( $T_{\mathrm{B}}, \mathrm{V}_0$ ) to the same final state $\left(T_{\mathrm{f}}, 5 V_0\right)$. The ratio of the specific heats at constant pressure and constant volume of this ideal gas is $\gamma$. What is the ratio $T_{\mathrm{A}} / T_{\mathrm{B}}$ ?View Solution
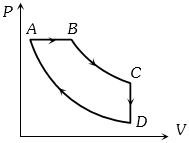
- 6The adjoining figure shows the $P-V$ diagram for a fixed mass of an ideal gas undergoing cyclic process. $AB$ represents isothermal process and $CA$ represents isochoric process.Which of the graph shown in the following figures represents the $P-T$ diagram of the cyclic process ?View Solution
- 7An ideal gas is subjected to an isothermal expansion such that its volume changes from $V_i$ to $V_f$ and pressure from $P_i$ to $P_f$. The work done on the gas is :View Solution
- 8Figure shows a polytropic process for an ideal gas. The work done by the gas will be in process $AB$ isView Solution
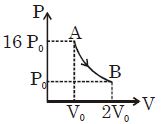
- 9The $PV$ diagram shows four different possible reversible processes performed on a monatomic ideal gas. Process $A$ is isobaric (constant pressure). Process $B$ is isothermal (constant temperature). Process $C$ is adiabatic. Process $D$ is isochoric (constant volume). For which process$(es)$ does the temperature of the gas decrease?View Solution
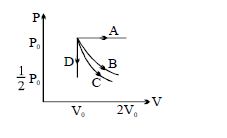
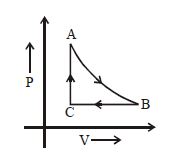
- 10A cyclic process $ABCD$ is shown in the figure $P-V$ diagram. Which of the following curves represent the same processView Solution