Let $A$ and $B$ the two gases and given : $\frac{{{T_A}}}{{{M_A}}} = 4.\frac{{{T_B}}}{{{M_B}}};$ where $T$ is the temperature and M is molecular mass. If ${C_A}$ and ${C_B}$ are the $r.m.s. $ speed, then the ratio $\frac{{{C_A}}}{{{C_B}}}$ will be equal to
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1$N\,moles$ of a diatomic gas in a cylinder are at a temperature $T$. Heat is supplied to the cylinder such that the temperature remains constant but $n\,moles$ of the diatomic gas get converted into monoatomic gas. What is the change in the total kinetic energy of the gas ?View Solution
- 2According to the kinetic theory of gases the $r.m.s.$ velocity of gas molecules is directly proportional toView Solution
- 3$Assertion :$ One mole of any substance at any temperature or volume always contains $6.02\times10^{23}$ molecules.View Solution
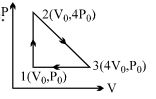
$Reason :$ One mole of a substance always refers to $S.T.P.$ conditions. - 4One mole of an ideal diatomic gas is taken through the cycle as shown in the figure.View Solution
$1 \rightarrow 2 :$ isochoric process
$2 \rightarrow 3 :$ straight line on $P-V$ diagram
$3 \rightarrow 1 :$ isobaric process
The average molecular speed of the gas in the states $1, 2$ and $3$ are in the ratio
- 5If three moles of monoatomic gas $\left(\gamma=\frac{5}{3}\right)$ is mixed with two moles of a diatomic gas $\left(\gamma=\frac{7}{5}\right)$, the value of adiabatic exponent $\gamma$ for the mixture is:View Solution
- 6The ratio of specific heats $(\gamma )$ of a ideal gas is given byView Solution
- 7At what temperature is the kinetic energy of a gas molecule double that of its value of $27°C$View Solution
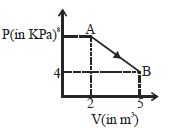
- 8A mole of an ideal diatomic gas undergoes a transition from $A$ to $B$ along a path $AB$ as shown in the figure, The change in internal energy of the gas during the transition is ...... $kJ$View Solution
- 9View SolutionSelect the incorrect relation. (Where symbols have their usual meanings)
- 10A gas in container $A$ is in thermal equilibrium with another gas in container $B.$ both contain equal masses of the two gases in the respective containers. Which of the following can be trueView Solution
