If pressure of a gas contained in a closed vessel is increased by $0.4\%$ when heated by $1°C,$ the initial temperature must be
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A mixture of $2\, moles$ of helium gas (atomic mass $= 4\, u$), and $1\, mole$ of argon gas (atomic mass $= 40\, u$) is kept at $300\, K$ in a container. The ratio of their rms speeds $\left[ {\frac{{{V_{rms}}{\rm{(helium)}}}}{{{V_{rms}}{\rm{(argon)}}}}} \right]$, is close toView Solution
- 2At $NTP$ the mass of one litre of air is $1.293\, gm.$ The value of specific gas constant will be ...... $J/K-gm$View Solution
- 3When a gas in a closed vessel was heated so as to increase its temperature by ${5^o}C$, there occurred an increase of $1\%$ in its pressure. The original temperature of the gas was ...... $^oC$View Solution
- 4According to $C.E.$ van der Waal, the interatomic potential varies with the average interatomic distance $(R)$ asView Solution
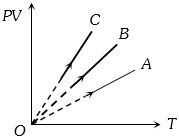
- 5$PV$ versus $T$ graph of equal masses of ${H_2}$, $He$ and ${O_2}$ is shown in fig. Choose the correct alternativeView Solution
- 6The capacity of a vessel is $3$ litres. It contains $6 \,gm$ oxygen, $8\, gm$ nitrogen and $5\, gm$ $C{O_2}$ mixture at $27°C.$ If $R = 8.31\, J/mole$ $ \times $ $kelvin,$ then the pressure in the vessel in $N/{m^2}$ will be (approx.)View Solution
- 7For one gram mol of a gas, the value of $R$ in the equation $PV = RT$ is nearly ...... $cal/K$View Solution
- 8The average kinetic energy of a helium atom at ${30^o}C$ isView Solution
- 9A cylinder of capacity $20$ litres is filled with ${H_2}$ gas. The total average kinetic energy of translatory motion of its molecules is $1.5 \times {10^5}\,J$. The pressure of hydrogen in the cylinder isView Solution
- 10Two non-reactive monoatomic ideal gases have their atomic masses in the ratio $2 : 3$. The ratio of their partial pressures, when enclosed in a vessel kept at a constant temperature, is $4 : 3$. The ratio of their densities is :-View Solution
