One litre of Helium gas at a pressure $76\, cm$ of $Hg$ and temperature ${27}^\circ$ is heated till its pressure and volume are doubled. The final temperature attained by the gas is ..... $^oC$
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionMolecules of a gas behave like
- 2An electron tube was sealed off during manufacture at a pressure of $1.2 \times {10^{ - 7}}$ $mm$ of mercury at $27°C.$ Its volume is $100\, cm^3$. The number of molecules that remain in the tube isView Solution
- 3View SolutionThe parameter that remains the same for molecules of all gases at a given temperature is :
- 4A $15\,g$ mass of nitrogen gas is enclosed in a vessel at a temperature $27\,^oC.$ Amount of heat transferred to the gas, so that rms velocity of molecules is doubled, is about ...... $kJ$ [Take $R = 8.3\,J/K\,mole$ ]View Solution
- 5At a certain temperature, the $r.m.s.$ velocity for ${O_2}$ is $400\, m/sec.$ At the same temperature, the $r.m.s.$ velocity for ${H_2}$ molecules will be ....... $m/sec$View Solution
- 6View SolutionWhich of the following statements about kinetic theory of gases is wrong
- 7For three low density gases $A, B, C$ pressure versus temperature graphs are plotted while keeping them at constant volume, as shown in the figure.View Solution
The temperature corresponding to the point ' $K$ ' is $...........\,{}^{\circ}\,C$
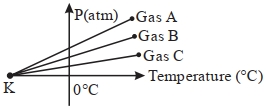
- 8If the oxygen ($O_2$) has root mean square velocity of $C\,m{s^{ - 1}}$, then root mean square velocity of the hydrogen ($H_2$) will beView Solution
- 9A vessel contains $16 \,g$ of hydrogen and $128 \,g$ of oxygen at standard temperature and pressure. The volume of the vessel in $cm ^{3}$ isView Solution
- 10A mixture of ideal gases $7\,\, kg$ of nitrogen and $11 \,\,kg$ of $CO_2$. ThenView Solution
(Take $\gamma$ for nitrogen and $CO_2$ as $1.4$ and $1.3$ respectively)
