If the oxygen ($O_2$) has root mean square velocity of $C\,m{s^{ - 1}}$, then root mean square velocity of the hydrogen ($H_2$) will be
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
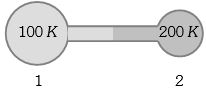
- 1Figure shows two flasks connected to each other. The volume of the flask $1$ is twice that of flask $2.$ The system is filled with an ideal gas at temperature $100\, K$ and $200 \,K $ respectively. If the mass of the gas in $1$ be $m$ then what is the mass of the gas in flask $2$View Solution
- 2The pressure and temperature of an ideal gas in a closed vessel are $720\, kPa$ and $40^oC$ respectively. If $\frac{1}{4}^{th}$ of the gas is released from the vessel and the temperature of the remaining gas is raised to $353^oC,$ the final pressure of the gas is ....... $kPa$View Solution
- 3A vessel contains $1$ mole of $O_2$ gas (molar mass $32$) at a temperature $T$. The pressure of the gas is $P$. An identical vessel containing one mole of $He$ gas (molar mass $4$) at a temperature $2T$ has a pressure ofView Solution
- 4Five molecules of a gas are having speeds $1,2,4,8$ and $16\,m/s$ at some instant. The root mean square velocity of the gas molecules is ..... $m/s$View Solution
- 5Five moles of helium are mixed with two moles of hydrogen to form a mixture. Take molar mass of helium $M_1=4\ g$ and that of hydrogen $M_2=2\ g$ If the internal energy of He sample of $100\,\,J$ and that of the hydrogen sample is $200\,\,J$, then the internal energy of the mixture is ..... $J$View Solution
- 6One mole of an ideal gas at $STP$ is heated in an insulated closed container until the average speed of its molecules is doubled. Its pressure would therefore increase by factor.View Solution
- 7View SolutionThat gas cannot be liquified
- 8Consider a sample of oxygen behaving like an ideal gas. At $300 \,K ,$ the ratio of root mean square (rms) velocity to the average velocity of gas molecule would be :View Solution
(Molecular weight of oxygen is $32 \,g / mol$ $\left. R =8.3 \,J K ^{-1} mol ^{-1}\right)$
- 9The temperature of an ideal gas is increased from $200\,K$ to $800\,K$. If r.m.s. speed of gas at $200\,K$ is $v_0$. Then, r.m.s. speed of the gas at $800\,K$ will be:View Solution
- 10One mole of an ideal gas passes through a process where pressure and volume obey the relation $P\, = {P_0}\,\left[ {1 - \frac{1}{2}{{\left( {\frac{{{V_0}}}{V}} \right)}^2}} \right]$. Here $P_0$ and $V_0$ are constants. Calculate the change in the temperature of the gas if its volume change from $V_0$ to $2V_0$View Solution
