A Container having $1$ mole of a gas at a temperature $27°C$ has a movable piston which maintains at constant pressure in container of $1 \,atm.$ The gas is compressed until temperature becomes $127°C$. The work done is ........ $J$ ($C_P$ for gas is $7.03\, cal/mol-K)$
Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1An ideal gas, initially in state $\left( P _{12}, V _1, T _1\right)$ is expanded isobarically to $\left( P _{12}, V _2, T _2\right)$, then adiabatically $\left( P _{34}, V _3, T _3\right)$. It is then contracted isobarically to $\left( P _{34}, V _4, T _4\right)$ and finally adiabatically back to the initial state. The efficiency of this cycle isView Solution
- 2One mole of an ideal gas is contained with in a cylinder by a frictionless piston and is initially at temperature $T$. The pressure of the gas is kept constant while it is heated and its volume doubles. If $R$ is molar gas constant, the work done by the gas in increasing its volume isView Solution
- 3A $P-T$ graph is shown for a cyclic process. Select correct statement regarding thisView Solution
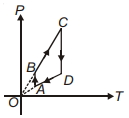
- 4Helium at ${27^o}C$ has a volume of $8$ litres. It is suddenly compressed to a volume of $1$ litre. The temperature of the gas will be ....... $^oC$ $[\gamma = 5/3]$View Solution
- 5The volume $V$ of a given mass of monoatomic gas changes with temperature $T$ according to the relation $V = KT ^{2 / 3}$. The workdone when temperature changes by $90\, K$ will be $x\,R$. The value of $x$ is $[ R =$ universal gas constant $]$View Solution
- 6An ideal refrigerator has a freezer at a temperature of $ - 13^\circ C.$ The coefficient of performance of the engine is $5.$ The temperature of the air (to which heat is rejected) will beView Solution
- 7A gas mixture consists of $8$ moles of argon and $6$ moles of oxygen at temperature $T$. Neglecting all vibrational modes, the total internal energy of the system isView Solution
- 8Heat given to a system is $35$ joules and work done by the system is $15$ joules. The change in the internal energy of the system will be ..... $J$View Solution
- 9The efficiency of an ideal heat engine working between the freezing point and boiling point of water, is ........ $\%$View Solution
- 10If heat given to a system is $6 \,kcal$ and work done is $ 6 kJ.$ Then change in internal energy is ....... $kJ$View Solution
