The temperature of sink of Carnot engine is ${27^o}C$. Efficiency of engine is $25\%.$ Then temperature of source is ...... $^oC$
Medium
(c) $\eta = 1 - \frac{{{T_2}}}{{{T_1}}}$ ==> $\frac{{25}}{{100}} = 1 - \frac{{300}}{{{T_1}}}$ ==> $\frac{1}{4} = 1 - \frac{{300}}{{{T_1}}}$
${T_1} = 400\,K = 127^\circ C$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionIf a cylinder containing a gas at high pressure explodes, the gas undergoes
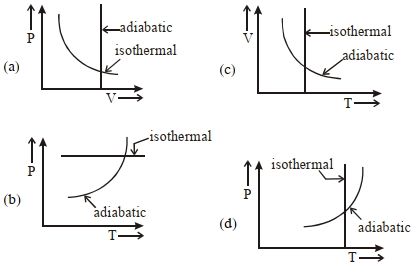
- 2View SolutionWhich one is the correct option for the two different thermodynamic processes ?
- 3An ideal gas has volume ${V_0}$ at ${27^o}C.$ It is heated at constant pressure so that its volume becomes $2{V_0}.$ The final temperature isView Solution
- 4View SolutionA reversible engine and an irreversible engine are working between the same temperatures. The efficiency of the ...........
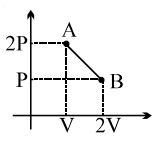
- 5The process $AB$ is shown in the diagram. As the gas is taken from $A$ to $B$, its temperatureView Solution
- 6Under isothermal condition, the pressure of a gas is given by $P = aV ^{-3}$, where $a$ is a constant and $V$ is the volume of the gas. The bulk modulus at constant temperature is equal to $..........\,P$View Solution
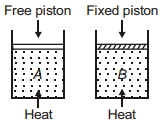
- 7Two cylinders contain same amount of ideal monatomic gas. Same amount of heat is given to two cylinders. If temperature rise in cylinder $A$ is $T_0$ then temperature rise in cylinder $B$ will be .........View Solution
- 8If $\Delta Q$ and $\Delta W$ represent the heat supplied to the system and the work done on the system respectively, then the first law of thermodynamics can be written asView Solution
- 9During an adiabatic process, the pressure of a gas is found to be proportional to the cube of its temperature. The ratio of $\frac{{{C_P}}}{{{C_V}}}$ for the gas isView Solution
- 10If $\gamma $ denotes the ratio of two specific heats of a gas, the ratio of slopes of adiabatic and isothermal $PV$ curves at their point of intersection isView Solution
