One mole of a monatomic ideal gas is taken through a cycle $ABCDA$ as shown in the $P-V$ diagram. Column $II$ gives the characteristics involved in the cycle. Match them with each of the processes qiven in Column $I$
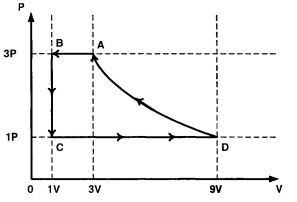
| Column $I$ | Column $II$ |
| $(A)$ Process $A \rightarrow B$ | $(p)$ Internal energy decreases. |
| $(B)$ Process $B \rightarrow C$ | $(q)$ Internal energy increases. |
| $(C)$ Process $C \rightarrow D$ | $(r)$ Heat is lost. |
| $(D)$ Process $D \rightarrow A$ | $(s)$ Heat is gained. |
| $(t)$ Work is done on the gas. |

IIT 2011, Diffcult
for $(A): A B$ is Isobaric compression
W is $- Ve ; \Delta U$ is $- Ve ; \Delta Q$ is - Ve
for $(B)$ : $C$ is Isochoric
$W = O ; \Delta U$ is $- Ve ; \Delta Q$ is - Ve
for $(C)$ : $CD$ is Isobaric expansion
$W$ is $+V e ; \Delta U$ is $+V e ; \Delta Q$ is $+V e$
for $(D)$ : $DA$ is Isothermal
W is $- Ve ; \Delta U=0 ; \Delta Q$ is - Ve
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1$Assertion :$ Reversible systems are difficult to find in real world.View Solution
$Reason :$ Most processes are dissipative in nature. - 2The efficiency of a Carnot engine operating with reservoir temperature of $100\,^{\circ} C$ and $-23\,^{\circ} C$ will beView Solution
- 3Gas obey $P^2V =$ constant. The initial temperature and volume are $T_0$ and $V_0$. If gas expands to volume $2V_0$, the final temp isView Solution
- 4The coefficient of performance of a Carnot refrigerator working between ${30^o}C$ and ${0^o}C$ isView Solution
- 5A system is provided with $200 \,cal$ of heat and the work done by the system on the surrounding is $40 \,J$. Then its internal energyView Solution
- 6A Carnot engine operates between ${227^o}C$ and ${27^o}C.$ Efficiency of the engine will beView Solution
- 7A source supplies heat to a system at the rate of $1000 \,W$. If the system performs work at a rate of $200\,W$. The rate at which internal energy of the system increases $.......\,W$View Solution
- 8A reversible cyclic process for an ideal gas is shown below. Here, $P , V$, and $T$ are pressure, volume and temperature, respectively. The thermodynamic parameters $q, w, H$ and $U$ are heat, work, enthalpy and internal energy, respectively.View Solution
The correct option ($s$) is (are)
$(A)$ $q_{A C}=\Delta U_{B C}$ and $W_{A B}=P_2\left(V_2-V_1\right)$ $(B)$ $W _{ BC }= P _2\left( V _2- V _1\right)$ and $q _{ BC }= H _{ AC }$ $(C)$ $\Delta H _{ CA }<\Delta U _{ CA }$ and $q _{ AC }=\Delta U _{ BC }$ $(D)$ $q_{B C}=\Delta H_{A C}$ and $\Delta H_{C A}>\Delta U_{C A}$
- 9If $\gamma $ denotes the ratio of two specific heats of a gas, the ratio of slopes of adiabatic and isothermal $PV$ curves at their point of intersection isView Solution
- 10View SolutionIn the following figures heat is absorbed by the gas
