[ $R$ is the gas constant]
$(1)$ Work done in this thermodynamic cycle $(1 \rightarrow 2 \rightarrow 3 \rightarrow 4 \rightarrow 1)$ is $| W |=\frac{1}{2} RT _0$
$(2)$ The ratio of heat transfer during processes $1 \rightarrow 2$ and $2 \rightarrow 3$ is $\left|\frac{ Q _{1 \rightarrow 2}}{ Q _{2 \rightarrow 3}}\right|=\frac{5}{3}$
$(3)$ The above thermodynamic cycle exhibits only isochoric and adiabatic processes.
$(4)$ The ratio of heat transfer during processes $1 \rightarrow 2$ and $3 \rightarrow 4$ is $\left|\frac{Q_{U \rightarrow 2}}{Q_{3 \rightarrow 4}}\right|=\frac{1}{2}$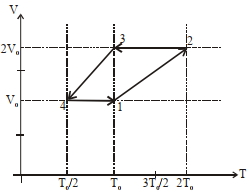
Process $2 \rightarrow 3$ is isochoric with $V =2 V _0$
Process $3 \rightarrow 4$ is isobaric with $P =\frac{ RT _0}{2 V _0}$
Process $4 \rightarrow 1$ is isochoric with $V = V _0$
Work in cycle $=\frac{ RT _0}{ V _0} \cdot V _0-\frac{ RT _0}{2 V _0} \cdot V _0=\frac{ RT _0}{2}$
$Q _{1-2}= nC _{ p } \Delta T = n \cdot \frac{5 R }{2} \cdot T _0$
$Q _{2-3}= nC _{ V } \Delta T = n \cdot \frac{3 R }{2} \cdot T _0$
$\therefore\left|\frac{ Q _{1-2}}{ Q _{2-3}}\right|=\frac{5}{3}$
$Q _{3-4}= nC _{ p } \Delta T = n \cdot \frac{5 R }{2} \cdot \frac{ T _0}{2}$
$\therefore\left|\frac{ Q _{1-2}}{ Q _{3-4}}\right|=2$
Ans.$1, 2$
Download our appand get started for free
Similar Questions
- 1A perfect gas of a given mass is heated first in a small vessel and then in a large vessel, such that their volumes remain unchanged. The $P-T$ curves areView Solution
- 2When the amount of work done is $333\, cal$ and change in internal energy is $167\, cal$, then the heat supplied is ....... $cal$View Solution
- 3In the figure given two processes $A$ and $B$ are shown by which a thermo-dynamical system goes from initial to final state $F.$ If $\Delta {Q_A}$ and $\Delta {Q_B}$ are respectively the heats supplied to the systems thenView Solution
- 4In an Isothermal change, the change in pressure and volume of a gas can be represented for three different temperature; $T _3 > T _2 > T _1$ as :View Solution
- 5An ideal heat engine exhausting heat at ${77^o}C$ is to have a $30\%$ efficiency. It must take heat at ...... $^oC$View Solution
- 6The temperature inside a refrigerator is $t_2 \,^o C$ and the room temperature is $t_1\,^o C.$ The amount of heat delivered to the room for each joule of electrical energy consumed ideally will beView Solution
- 7Which of the accompanying $PV$, diagrams best represents an isothermal processView Solution
- 8Two gases of equal mass are in thermal equilibrium. If ${P_a},\,{P_b}$ and ${V_a}$ and ${V_b}$ are their respective pressures and volumes, then which relation is trueView Solution
- 9The isothermal Bulk modulus of an ideal gas at pressure $P$ isView Solution
- 10Two samples $A$ and $B$ of a gas initially at the same pressure and temperature are compressed from volume $ V$ to $ V/2$ ($A$ isothermally and adiabatically). The final pressure of $ A$ isView Solution
