A real gas behaves like an ideal gas if its
IIT 2010, Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionMolecules of a gas behave like
- 2View SolutionThe relation between two specific heats of a gas is
- 3An ideal gas is initially at temperature $T$ and volume $V.$ Its volume is increased by $\Delta V$ due to an increase in temperature $\Delta T,$ pressure remaining constant. The quantity $\delta = \Delta V/(V\Delta T)$ varies with temperature asView Solution
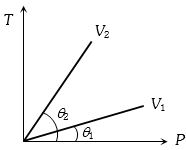
- 4From the following $P-T$ graph what interference can be drawnView Solution
- 5An ideal gas is enclosed in a container of volume $V$ at a pressure $P$. It is being pumped out of the container by using a pump with stroke volume $v$. What is final pressure in container after $n$-stroke of the pump? (assume temperature remains same)View Solution
- 6What is the value of $\frac{R}{{{C_P}}}$ for diatomic gasView Solution
- 7Two non-reactive monoatomic ideal gases have their atomic masses in the ratio $2 : 3$. The ratio of their partial pressures, when enclosed in a vessel kept at a constant temperature, is $4 : 3$. The ratio of their densities is :-View Solution
- 8A sample of an ideal gas occupies a volume $V$ at a pressure $P$ and absolute temperature $T,$ the mass of each molecule is $m.$ The expression for the density of gas is ($k =$ Boltzmann’s constant)View Solution
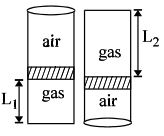
- 9An ideal gas is trapped inside a test tube of cross-sectional area $20 \times 10^{-6} \,\,m^2$ as shown in the figure. The gas occupies a height $L_1$ at the bottom of the tube and is separated from air at atmospheric pressure by a mercury column of mass $0.002\,\, kg$. If the tube is quickly turned isothermally, upside down so that $L_2$ mercury column encloses the gas from below. The gas now occupies height $L_1$ in the tube. The ratio $L_1$ is [Take atmospheric pressure $= 10^5 Nm^{-2}]$View Solution
- 10View SolutionKinetic theory of gases provide a base for
