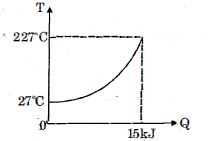
$Q$ amount of heat is given to $0.5\ mole$ of an ide al mono-atomic gas by a process $TV^n$ constant. Following graph shows variation of temperature with $Q$ . Find value of $n$.

Advanced
$\Delta \mathrm{Q}=\Delta \mathrm{U}+\Delta \mathrm{W}$
$\Delta \mathrm{Q}=\frac{\mathrm{f}}{2} \mathrm{n} \mathrm{R} \Delta \mathrm{T}+\frac{\mathrm{n} \mathrm{R} \Delta \mathrm{T}}{1-\mathrm{n}_{0}}$
$15 \times 10^{3}=\frac{3}{2} \times \frac{1}{2} \times \frac{25}{3} \times 200+\frac{0.5 \times 25 / 3 \times 200}{1-\mathrm{n}_{0}}$
$150=\frac{25}{2}+\frac{25}{3\left(1-n_{0}\right)}$
$6=\frac{1}{2}+\frac{1}{3\left(1-n_{0}\right)}$
$5.5=\frac{1}{3\left(1-n_{0}\right)}$
$16.5-16.5 \mathrm{n}_{0}=1$
$-16.5 \mathrm{n}_{0}=-15.5$
$\mathrm{T} \mathrm{V}_{0}^{\mathrm{n}-1}=\mathrm{constant}$
$\mathrm{n}_{0}-1=\mathrm{n}$
$\frac{{155}}{{165}} - 1 = n$
$\frac{-10}{165}=n$
$n=\left(\frac{-2}{33}\right)$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1For an ideal heat engine, the temperature of the source is $127\,^{\circ} C$. In order to have $60\, \%$ efficiency the temperature of the sink should be $........\,{ }^{\circ} C$. (Round off to the Nearest Integer)View Solution
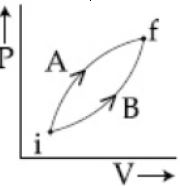
- 2Following figure shows two processes $A$ and $B$ for a gas. If $\Delta Q_A$ and $\Delta Q_B$ are the amount of heat absorbed by the system in two cases, and $\Delta U_A$ and $\Delta U_B$ are changes in internal energies, respectively, thenView Solution
- 3View SolutionWhich statement is incorrect?
- 4View SolutionAt constant pressure how much fraction of heat supplied to gas is converted into mechanical work?
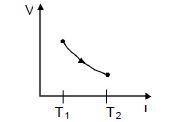
- 5One mole of an ideal gas undergoes a cyclic process, consisting of two isochores and two isobars. Temperature at $1$ and $3$ equal to $T_1$ and $T_3$ respectively. The work done by the gas over the cycle, if the point $2$ and $4$ lie on the same isothermView Solution
- 6The figure shows a process $AB$ undergone by $2$ moles of an ideal diatomic gas. The process $AB$ is in such a way that $VT =$ constant. $T_1 = 300 K $and $T_2 = 500 K$ ( $R = $ gas constant)View Solution
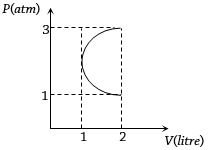
- 7In the $P-V$ diagram shown in figure $ABC$ is a semicircle. The work done in the process $ABC$ isView Solution
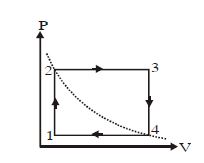
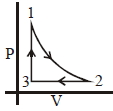
- 8Which of the following is an equivalent cyclic process corresponding to the thermodynamic cyclic given in the figure? where, $1 \rightarrow 2$ is adiabatic.View Solution
(Graphs are schematic and are not to scale)
- 9Two kg of water is converted into steam by boiling at atmospheric pressure. The volume changes from $2 \times {10^{ - 3}}\,{m^3}$ to $3.34{m^3}.$ The work done by the system is about ....... $kJ$View Solution
- 10In an adiabatic process, the density of a diatomic gas becomes $32$ times its initial value. The final pressure of the gas is found to be $n$ times the initial pressure. The value of $n$ isView Solution
