Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionWhich of the following species show maximum volatility?
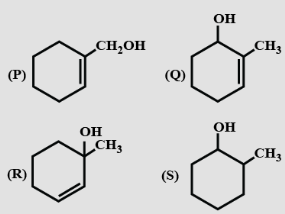
- 2View SolutionDehydration of the following in increasing order is:
- 3The compound $A$ on treatment with $Na$ gives $B,$ and with $\ce{PCI_{5 }}$ gives $C. B$ and $C$ react together to give diethyl ether. $A, B$ and $C$ are in the order.View Solution
- 4$\ce{IUPAC}$ name of $m-$cresol is $.......$View Solution
- 5The correct decreasing order of dehydration of following alcohols with conc. $\ce{H_{2}SO_{4}}$ is$:$View Solution
- 6View SolutionMethanol is now prepared from:
- 7View SolutionWhich compound is the most soluble in water?
- 8View SolutionWhich of the following compounds will react with sodium hydroxide solution in water?
- 9Compound $\ce{C_2H_6O}$ has two isomers $X$ and $Y.$ On reaction with $HI, X$ gives alkyl iodide and water while $Y$ gives alkyl iodide and alcohol.Compounds $X$ and $Y$ are respectively:View Solution
- 10View SolutionThe miscibility of ether with _______ resembles those of alcohol of the same molecular mass.