The adiabatic Bulk modulus of a diatomic gas at atmospheric pressure is
Diffcult
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1An iron rod of heat capacity $C$ is heated to temperature $8T_0$ . It is then put in a cylindrical vessel of adiabatic walls having two moles of air which can be treated as diatomic ideal gas at temperature $T_0$ and closed by a movable piston which is also adiabatic. The atmospheric pressure is $P_0$ . The cylinder with the piston combined have heat capacity $2C$ . Find the equilibrium temperature . (Assume temperature of air to be uniform and equal to vessel at all times) .View Solution
- 2Two moles of helium gas are taken over the cycle $ABCDA$, as shown in the $P-T$ diagram. The net work done on the gas in the cycle $ABCDA$ is ...... $R$View Solution

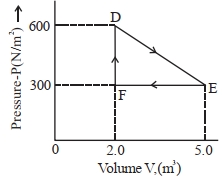
- 3A thermodynamic system is taken from an original state $D$ to an intermediate state $E$ by the linear process shown in the figure. Its volume is then reduced to the original volume from $E$ to $F$ by an isobaric process. The total work done by the gas from $D$ to $E$ to $F$ will be $......J$View Solution
- 4Under isothermal condition, the pressure of a gas is given by $P = aV ^{-3}$, where $a$ is a constant and $V$ is the volume of the gas. The bulk modulus at constant temperature is equal to $..........\,P$View Solution
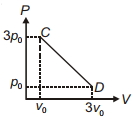
- 5The process $C D$ is shown in the diagram. As system is taken from $C$ to $D$, what happens to the temperature of the system?View Solution
- 6View SolutionThe process in which no heat enters or leaves the system is termed as
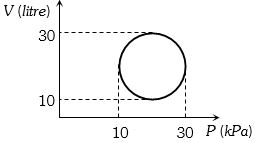
- 7View SolutionHeat energy absorbed by a system in going through a cyclic process shown in figure is
- 8An engineer claims to have made an engine delivering $10 kW$ power with fuel consumption of $1 g/sec$. The calorific value of the fuel is $2 kcal/g$. Is the claim of the engineerView Solution
- 9A frictionless heat engine can be $100 \%$ efficient only if its exhaust temperature is ............View Solution
- 10One mole of an ideal gas $(\gamma = 1.4)$ is adiabatically compressed so that its temperature rises from $27\,^oC$ to $35\,^oC$. The change in the internal energy of the gas is .... $J$ (given $R = 8.3 \,J/mole/K$)View Solution
