The amount of heat energy required to raise the temperature of $1\, g$ of Helium at $NTP,$ from $T_1 K$ to $T_2 K$ is
AIPMT 2013, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1The temperature of an ideal gas is increased from $27°C$ to $927°C.$ The root mean square speed of its molecules becomesView Solution
- 2$5$ moles of oxygen is heated at constant volume from $10°C$ to $20°C.$ The change in the internal energy of the gas is (the gram molecular specific heat of oxygen at constant pressure...... $cal$ ${C_p} = 8$ $cal/mole \,°C$ and $R = 8.3 cal/mole\, °C)$View Solution
- 3View SolutionA diatomic molecule has how many degrees of freedom
- 4A gas mixture consists of $2$ moles of oxygen and 4 moles of neon at temperature $T$. Neglecting all vibrational modes, the total internal energy of the system will be $...........\,RT$View Solution
- 5A flat plate is moving normal to its plane through a gas under the action of constant force $F$. The gas is kept at a very low pressure. The speed of the plate $v$ is much less than the average speed $u$ of the gas molecules. Which of the following options is/are true?View Solution
[$A$] The resistive force experienced by the plate is proportional to $\mathrm{v}$
[$B$] The pressure difference between the leading and trailing faces of the plate is proportional to uv
[$C$] The plate will continue to move with constant non-zero acceleration, at all times
[$D$] At a later time the external force $F$ balances the resistive force.
- 6One mole of an ideal gas at $STP$ is heated in an insulated closed container until the average speed of its molecules is doubled. Its pressure would therefore increase by factor.View Solution
- 7When unit mass of water boils to become steam at $100\,^0C$, it absorbs $Q$ amount of heat. The densities of water and steam at $100\,^0C$ are $\rho_1$ and $\rho_2$ respectively and the atmospheric pressure is $p_0$. The increase in internal energy of the water isView Solution
- 8The value of critical temperature in terms of Vander Waal’s constant $a$ and $b$ isView Solution
- 9An ideal gas is trapped inside a test tube of cross-sectional area $20 \times 10^{-6} \,\,m^2$ as shown in the figure. The gas occupies a height $L_1$ at the bottom of the tube and is separated from air at atmospheric pressure by a mercury column of mass $0.002\,\, kg$. If the tube is quickly turned isothermally, upside down so that $L_2$ mercury column encloses the gas from below. The gas now occupies height $L_1$ in the tube. The ratio $L_1$ is [Take atmospheric pressure $= 10^5 Nm^{-2}]$View Solution
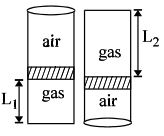
- 10View SolutionOn absolute temperature, the kinetic energy of the molecules
