The value of critical temperature in terms of Vander Waal’s constant $a$ and $b$ is
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1The molar heat capacity in a process of a diatomic gas if it does a work of $\frac{Q}{4}$ when a heat of $Q$ is supplied to it isView Solution
- 2Let $A$ and $B$ the two gases and given : $\frac{{{T_A}}}{{{M_A}}} = 4.\frac{{{T_B}}}{{{M_B}}};$ where $T$ is the temperature and M is molecular mass. If ${C_A}$ and ${C_B}$ are the $r.m.s. $ speed, then the ratio $\frac{{{C_A}}}{{{C_B}}}$ will be equal toView Solution
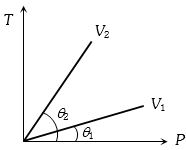
- 3From the following $P-T$ graph what interference can be drawnView Solution
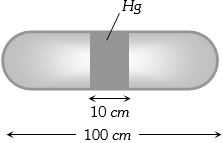
- 4A horizontal uniform glass tube of $100 \,cm$, length sealed at both ends contain $10 \,cm$ mercury column in the middle. The temperature and pressure of air on either side of mercury column are respectively $81°C$ and $76\, cm$ of mercury. If the air column at one end is kept at $0°C$ and the other end at $273°C$, the pressure of air which is at $0°C$ is (in $cm$ of $Hg$)View Solution
- 5The molar specific heats of an ideal gas at constant pressure and volume are denoted by $C _{ P }$ and $C _{ V }$ respectively. If $\gamma=\frac{C_{p}}{C_{V}}$ and $R$ is the universal gas constant, then $C _{ V }$ is equal toView Solution
- 6View SolutionAn ideal gas is filled in a vessel, then
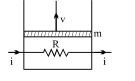
- 7A resistance coil connected to an external battery is placed inside an adiabatic cylinder fitted with a frictionless pistn and containing an ideal gas. A current $i$ flows through the coil which has a resistance $R$. At what speed must the piston move upward in order that the temperature of the gas remains uchanged? Neglect atmospheric pressure.View Solution
- 8View SolutionFor ideal gas, which statement is not true
- 9View SolutionThree vessels of equal capacity have gases at the same temperature and pressure. The first vessel contains helium (monoatomic), the second contains fluorine (diatomic) and the third contains sulfur hexafluoride (polyatomic). The correct statement, among the following is
- 10Abarometer tube, containing mercury, is lowered in a vessel containing mercury until only $50\,\, cm$ of the tube is above the level of mercury in the vessel. If the atmospheric pressure is $75 \,\,cm$ of mercury, what is the pressure at the top of the tube?View Solution
