The bond angle of the bond $C−O−C$ in methoxy methane is:
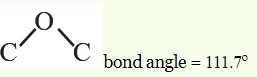
Ethers feature $C–O–C$ linkage defined by a bond angle of about $110^\circ$ and $C–O$ bond length of about $140\ pm.$
In methoxymethane, the bond angle is $111.7^\circ $. Methoxymethane show positive deviation in the bond angle. This is basically due to the steric crowding on carbon atoms of methyl group.
Due to the crowding, the repulsion between bond pair of carbons and lone pair of oxygen is low. Thus, the bond angle of methoxymethane is $111.7^\circ.$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionWhich of the following alcohol on dehydration will produce a pair of isomeric alkene?
- 2View SolutionMolecular formula of ethanol is?
- 3The best method to prepare $3-$methylbutan$-2-ol$ from $3-$methylbut$-1-$ene is:View Solution
- 4View SolutionThe correct order of boiling point for primary (1), secondary (2) and tertiary (3) alcohols is:
- 5View SolutionWhich of the following alkenes on acid-catalysed hydration gives a tertiary alcohol:
- 6View SolutionThe decreasing order of boiling points of the following alcohols is?
- 7View SolutionGive the IUPAC name of the above compound.
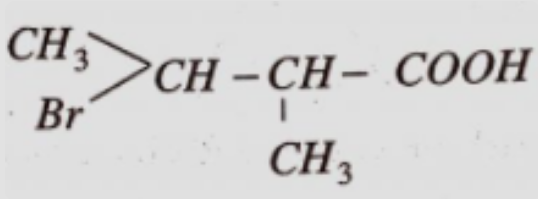
- 8View SolutionPhenol reacts with alkali to give:
- 9View SolutionLower alcohols are highly soluble in water due to _____.
- 10Carbinol is a trivial name of$:$View Solution
