$\eta = \left( {1 - \frac{{{T_2}}}{{{T_1}}}} \right)$
Freezing point of water $ = {0^ \circ }C = 273\,K$
Boiling point of water$ = {100^ \circ }C = \left( {100 + 273} \right)K$
$ = 373\,K$
$T_2$ Sink temperature$=273 K$
$T_1$ Source temperature $=373 K$
$\% \eta = \left( {1 - \frac{{{T_2}}}{{{T_1}}}} \right) \times 100 = \left( {1 - \frac{{273}}{{373}}} \right) \times 100$
$ = \left( {\frac{{100}}{{373}}} \right) \times 100 = 26.8\% $
Download our appand get started for free
Similar Questions
- 1The efficiency of a Carnot engine operating with reservoir temperature of $100\,^{\circ} C$ and $-23\,^{\circ} C$ will beView Solution
- 2$0.08 \mathrm{~kg}$ air is heated at constant volume through $5^{\circ} \mathrm{C}$. The specific heat of air at constant volume is $0.17 \mathrm{kcal} / \mathrm{kg}^{\circ} \mathrm{C}$ and $\mathrm{J}=4.18$ joule $/ \mathrm{cal}$. The change in its internal energy is approximately.View Solution
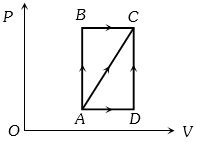
- 3A thermodynamic process is shown in the figure. The pressures and volumes corresponding to some points in the figure are :View Solution
${P_A} = 3 \times {10^4}Pa,\;{P_B} = 8 \times {10^4}Pa$ and ${V_A} = 2 \times {10^{ - 3}}{m^3},\;{V_D} = 5 \times {10^{ - 3}}{m^3}$
In process $AB$, $600 J$ of heat is added to the system and in process $BC, 200 J $ of heat is added to the system. The change in internal energy of the system in process $ AC$ would be ...... $J$
- 4Half mole of an ideal monoatomic gas is heated at constant pressure of $1\, atm$ from $20\,^oC$ to $90\,^oC$. Work done by has is close to ..... $J$ (Gas constant $R = 8.31\, J/mol.K$)View Solution
- 5A diatomic gas, having $C_{p}=\frac{7}{2} R$ and $C _{ v }=\frac{5}{2} R ,$ is heated at constant pressure. The ratio $dU : dQ : dW :$View Solution
- 6The heat capacity of one mole an ideal is found to be $C_V=3 R(1+a R T) / 2$, where $a$ is a constant. The equation obeyed by this gas during a reversible adiabatic expansion isView Solution
- 7View SolutionIn an isothermal change, an ideal gas obeys
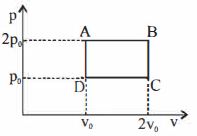
- 8The above $P-V$ diagram represents the thermodynamic cycle of an engine, operating with an ideal monatomic gas. The amount of heat, extracted from the source in a single cycle isView Solution
- 9In an $H_2$ gas process, $PV^2 =$ constant. The ratio of work done by gas to change in its internal energy isView Solution
- 10Even Carnot engine cannot give $100\%$ efficiency because we cannotView Solution
