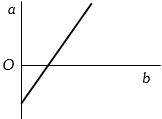
The expansion of unit mass of a perfect gas at constant pressure is shown in the diagram. Here

Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A flat plate is moving normal to its plane through a gas under the action of constant force $F$. The gas is kept at a very low pressure. The speed of the plate $v$ is much less than the average speed $u$ of the gas molecules. Which of the following options is/are true?View Solution
[$A$] The resistive force experienced by the plate is proportional to $\mathrm{v}$
[$B$] The pressure difference between the leading and trailing faces of the plate is proportional to uv
[$C$] The plate will continue to move with constant non-zero acceleration, at all times
[$D$] At a later time the external force $F$ balances the resistive force.
- 2An air bubble of volume $1.0\; cm ^{3}$ rises from the bottom of a lake $40\; m$ deep at a temperature of $12\,^{\circ} C$. To what volume (in $cm^3$) does it grow when it reaches the surface, which is at a temperature of $35\,^{\circ} C ?$View Solution
- 3$P-V$ diagram of a diatomic gas is a straight line passing through origin. The molar heat capacity of the gas in the process will beView Solution
- 4The translational degrees of freedom $(f)$ and rotational degrees of freedom $(f)$ of $\mathrm{CH}_4$ molecule are:View Solution
- 5A container contains $32 \,g$ of $O _2$ at a temperature $T$. The pressure of the gas is $P$. An identical container containing $4 g$ of $H _2$ at a temperature $2 T$ has a pressure of ............View Solution
- 6Modern vacuum pumps can evacuate a vessel down to a pressure of $4.0 \times {10^{ - 15}}\, atm$ at room temperature $(300\, K)$. Taking $R = 8.0\, JK^{-1}\, mole^{-1}$ , $1\, atm = 10^5\, Pa$ and $N_ {Avogadro} = 6 \times 10^{23}\, mole^{-1}$ , the mean distance between molecules of gas in an evacuated vessel will be of the order ofView Solution
- 7View SolutionWhen air is filled in the balloon, the pressure and volume both increases while temperature does not change. Here Boyle's law is not obeyed because
- 8If the degree of freedom of a gas are $f,$ then the ratio of two specific heats ${C_P}/{C_V}$ is given byView Solution
- 9An electron tube was sealed off during manufacture at a pressure of $1.2 \times {10^{ - 7}}$ $mm$ of mercury at $27°C.$ Its volume is $100\, cm^3$. The number of molecules that remain in the tube isView Solution
- 10If $\alpha$ moles of a monoatomic gas are mixed with $\beta$ moles of a polyatomic gas and mixture behaves like diatomic gas, then [neglect the vibrational mode of freedom]View Solution
