$Assertion :$ In an isolated system the entropy increases.
$Reason :$ The processes in an isolated system are adiabatic.
$Reason :$ The processes in an isolated system are adiabatic.
AIIMS 2006, Easy
In an unisolated system, heat may enter into or escape from the system due to which entropy may increase or decrease but for isolated system we do not consider exchange of heat, so, in this case entropy will always increase as the process is spontaneous. An adiabatic process involves no exchange of heat. We also define isolated system as having no exchange of heat with the surrounding so it process in an isolated system are adibatic. The two statements are independently correct but not co-related.
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
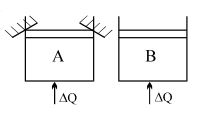
- 1Two identical vessels $A \& B$ contain equal amount of ideal monoatomic gas. The piston of $A$ is fixed but that of $B$ is free. Same amount of heat is absorbed by$A \& B$. If $B'$s internal energy increases by $100 \,\,J$ the change in internal energy of $A$ is ...... .$J$View Solution
- 2The specific heat at constant pressure of a real gas obeying $\mathrm{PV}^2=\mathrm{RT}$ equation is :View Solution
- 3If a Carnot’s engine functions at source temperature $127^o C$ and at sink temperature $87^o C$, what is its efficiency ....... $\%$View Solution
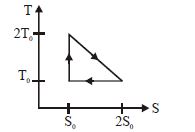
- 4View SolutionThe temperature-entropy diagram of a reversible engine cycle is given in the figure. Its efficiency is
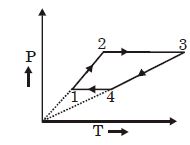
- 5Three moles of an ideal monoatomic gas perform a cycle as shown in the figure. The gas temperature in different states are: $T_1 = 400\, K, T_2 = 800\, K, T_3 = 2400\,K$ and $T_4 = 1200\,K.$ The work done by the gas during the cycle is ........ $kJ$View Solution
- 6In a mechanical refrigerator, the low temperature coils are at a temperature of $-23°C$ and the compressed gas in the condenser has a temperature of $27°C.$ The theoretical coefficient of performance isView Solution
- 7The pressure and volume of an ideal gas are related as $\mathrm{PV}^{3 / 2}=\mathrm{K}$ (Constant). The work done when the gas is taken from state $A\left(P_1, V_1, T_1\right)$ to state $\mathrm{B}\left(\mathrm{P}_2, \mathrm{~V}_2, \mathrm{~T}_2\right)$ is :View Solution
- 8$200\,g$ water is heated from $40\,^oC$ to $60\,^oC.$ Ignoring the slight expansion of water, the change in its internal energy is close to ...... $kJ$ (Given specific heat of water $=4184\,J/kgK$ )View Solution
- 9View SolutionA cycle tyre bursts suddenly. This represents an
- 10Choose the incorrect statement from the followingView Solution
$S1:$ The efficiency of a heat engine can be $1,$ but the coefficient of performance of a refrigerator can never be infinity.
$S2:$ The first law of thermodynamics is basically the principle of conservation of energy.
$S3:$ The second law of thermodynamics does not allow several phenomena consistent with the first law.
$S4:$ A process,whose only result is to transfer heat from a colder to a hotter object, is impossible.
