For a certain process, pressure of diatomic gas varies according to the relation $P = aV^2$, where $a$ is constant. What is the molar heat capacity of the gas for this process?
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1When a gas filled in a closed vessel is heated by raising the temperature by $1^{\circ} C$, its pressure increase by $0.4 \%$. The initial temperature of the gas is ..........$K$View Solution
- 2View SolutionThe expansion of unit mass of a perfect gas at constant pressure is shown in the diagram. Here
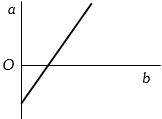
- 3View SolutionYou have two containers of equal volume. One is full of helium gas. The other holds an equal mass of nitrogen gas. Both gases have the same pressure. How does the temperature of the helium compare to the temperature of the nitrogen ?
- 4View SolutionWhich of the following graphs represent the behavior of an ideal gas ? Symbols have their usual meaning.
- 5View SolutionThe r.m.s. speed of gas molecules is given by
- 6View SolutionIf hydrogen gas is heated to a very high temperature, then the fraction of energy possessed by gas molecules correspond to rotational motion ...........
- 7If pressure of $C{O_2}$ (real gas) in a container is given by $P = \frac{{RT}}{{2V - b}} - \frac{a}{{4{b^2}}}$ then mass of the gas in container is ...... $gm$View Solution
- 8The change in the magnitude of the volume of an ideal gas when a small additional pressure $\Delta P$ is applied at a constant temperature, is the same as the change when the temperature is reduced by a small quantity $\Delta T$ at constant pressure. The initial temperature and pressure of the gas were $300\, K$ and $2\; atm$ respectively. If $|\Delta T|=C|\Delta P|$ then value of $C$ in $(K / a t m)$ is......View Solution
- 9The $r.m.s.$ velocity will be greater forView Solution
- 10The molar specific heat of a gas as given from the kinetic theory is $\frac{5}{2} R$. If it is not specified whether it is $C _{ P }$ or $C _{ V }$, one could conclude that the molecules of the gasView Solution
