The kinetic energy of one gram molecule of a gas at normal temperature and pressure is $(R = 8.31\,J/mole - K)$
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Five particles have speeds $1, 2, 3, 4, 5 \,\,m/s$. the average velocity of the particles is (in $m/s$)View Solution
- 2View SolutionWhich of the following plots represents schematically the dependence of the time period of a pendulum, if measured and plotted as a function of the amplitude of its oscillations? (Note amplitude need not be small)
- 3A diatomic gas of molecules weight $30\,\, gm/mole$ is filled in a container at $27\,^oC$. It is moving at a velocity $100\,\, m/s$. If it is suddenly stopped, the rise in temperature of gas is :View Solution
- 4If the ratio of vapour density for hydrogen and oxygen is $\frac{1}{{16}}$, then under constant pressure the ratio of their rms velocities will beView Solution
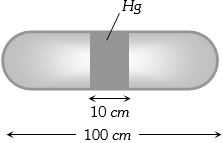
- 5A horizontal uniform glass tube of $100 \,cm$, length sealed at both ends contain $10 \,cm$ mercury column in the middle. The temperature and pressure of air on either side of mercury column are respectively $81°C$ and $76\, cm$ of mercury. If the air column at one end is kept at $0°C$ and the other end at $273°C$, the pressure of air which is at $0°C$ is (in $cm$ of $Hg$)View Solution
- 6A pressure cooker contains air at $1$ atm and $30^o C$. If the safety value of the cooler blows when the inside pressure $ \ge 3$ atm, then the maximum temperature of the air, inside the cooker can be .... $^oC$View Solution
- 7At ..... $K$ temperature average speed of $O_2$ will be ${\left( {\frac{7}{4}} \right)^{th}}$ of rms speed of $N_2$ at $47\,^oC$View Solution
- 8The volume of a gas at $20°C$ is $200\, ml$. If the temperature is reduced to $-20°C$ at constant pressure, its volume will be ...... $ml$View Solution
- 9If the intermolecular forces vanish away, the volume occupied by the molecules contained in $4.5 \,kg$ water at standard temperature and pressure will beView Solution
- 10The temperature of an ideal gas is increased from $200\,K$ to $800\,K$. If r.m.s. speed of gas at $200\,K$ is $v_0$. Then, r.m.s. speed of the gas at $800\,K$ will be:View Solution
