The volume of a gas at pressure $21 \times {10^4}\,N/{m^2}$ and temperature $27^o C$ is $83 $ litres. If $R = 8.3\ J/mol/K$, then the quantity of gas in $gm-mole$ will be
Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1The Mean Free Path $\ell$ for a gas molecule depends upon diameter, $d$ of the molecule asView Solution
- 2A hot air balloon with a payload rises in the air. Assume that the balloon is spherical in shape with diameter of $11.7 \,m$ and the mass of the balloon and the payload (without the hot air inside) is $210 \,kg$. Temperature and pressure of outside air are $27^{\circ} C$ and $1 atm =10^5 \,N / m ^2$, respectively. Molar mass of dry air is $30 \,g$. The temperature of the hot air inside is close to .......... $^{\circ} C$ [The gas constant, $R=8.31 \,JK ^{-1} mol ^{-1}$ ]View Solution
- 3A thermally insulated vessel contains an ideal gas of molecular mass $M$ and ratio of specific heats $1.4$. Vessel is moving with speed $v$ and is suddenly brought to rest. Assuming no heat is lost to the surrounding and vessel temperature of the gas increases by ... ( $R =$ universal gas constant )View Solution
- 4The diameter of oxygen molecule is $2.94 \times {10^{ - 10}}m.$ The Vander Waal’s gas constant ‘$b’$ in ${m^3}/mol$ will beView Solution
- 5Five moles of helium are mixed with two moles of hydrogen to form a mixture. Take molar mass of helium $M_1=4\ g$ and that of hydrogen $M_2=2\ g$ The equivalent molar mass of the mixture isView Solution
- 6View SolutionIndicator diagram is a ...........
- 7Two thermodynamical process are shown in the figure. The molar heat capacity for process $A$ and $B$ are $C_A$ and $C_B$. The molar heat capacity at constant pressure and constant volume are represented by $C_P$ and $C_V$, respectively. Choose the correct statement.View Solution
- 8View SolutionThe molecules of an ideal gas at a certain temperature have
- 9Figure shows the isotherms of a fixed mass of an ideal gas at three temperatures $T_A, T_B$ and $T_C$, thenView Solution
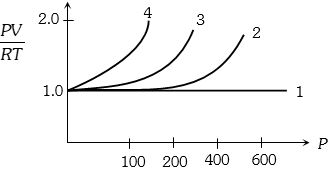
- 10A fix amount of nitrogen gas ($1$ mole) is taken and is subjected to pressure and temperature variation. The experiment is performed at high pressure as well as high temperatures. The results obtained are shown in the figures. The correct variation of $PV/RT$ with $P$ will be exhibited byView Solution