The only possibility of heat flow in a thermos flask is through its cork which is $75 cm^2$ in area and $5 cm$ thick. Its thermal conductivity is $0.0075 cal/cmsec^oC$. The outside temperature is$ 40^oC$ and latent heat of ice is $80 cal g^{-1}$. Time taken by $500 g$ of ice at $0^oC$ in the flask to melt into water at $0^oC$ is ....... $hr$

Diffcult
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionWhile measuring the thermal conductivity of a liquid, we keep the upper part hot and lower part cool, so that
- 2View SolutionIn which of the following process, convection does not take place primarily
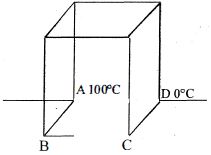
- 3Find Temperature difference between $B$ and $C$ ? (All rods are identical)View Solution
- 4A hollow copper sphere $S$ and a hollow copper cube $ C$ , both of negligible thin walls of same area, are filled with water at $90°C$ and allowed to cool in the same environment. The graph that correctly represents their cooling isView Solution
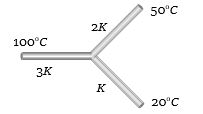
- 5Three rods of the same dimension have thermal conductivities $3K$ , $2K$ and $K$ . They are arranged as shown in fig. Given below, with their ends at $100^oC, 50^oC $and $20^oC$. The temperature of their junction is ......... $^oC$View Solution
- 6A slab consists of two parallel layers of two different materials of same thickness having thermal conductivities $K_1$ and $K_2$ . The equivalent conductivity of the combination isView Solution
- 7The lengths and radii of two rods made of same material are in the ratios $1 : 2$ and $2 : 3$ respectively. If the temperature difference between the ends for the two rods be the same, then in the steady state, the amount of heat flowing per second through them will be in the ratioView Solution
- 8The temperature of hot and cold end of a $20cm$ long rod in thermal steady state are at ${100^o}C$ and ${20^o}C$ respectively. Temperature at the centre of the rod is...... $^oC$View Solution
- 9One end of a copper rod of length $1.0\;m$ and area of cross-section ${10^{ - 3}}$ is immersed in boiling water and the other end in ice. If the coefficient of thermal conductivity of copper is $92\;cal/m{\rm{ - }}s{{\rm{ - }}^o}C$ and the latent heat of ice is $8 \times {10^4}cal/kg$, then the amount of ice which will melt in one minute isView Solution
- 10A body cools from ${60^o}C$ to ${50^o}C$ in $10$ minutes when kept in air at ${30^o}C$. In the next $10$ minutes its temperature will beView Solution
