A reversible engine and an irreversible engine are working between the same temperatures. The efficiency of the ...........
Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A gas is enclosed in a cylinder with a movable frictionless piston. Its initikl thermodynamic state at pressure $P_i=10^5 \mathrm{~Pa}$ and volume $V_i=10^{-3} \mathrm{~m}^3$ chanıes to i final state at $P_f=(1 / 32) \times 10^5 \mathrm{~Pa}$ and $V_f=8 \times 10^{-3} \mathrm{~m}^3$ in an adiabatic quasi-static process, such that $P^3 V^5=$ constant. Consider another thermodynamic process that brings the system from the same initial state to the same final state in two steps: an isobaric expansion at $P$, followed by an isochoric (isovolumetric) process at volume $V_f$. The amount of heat supplied to the system in the two-step process is approximatelyView Solution
- 2An ideal gas is taken through a quasi-static process described by $P = \alpha\, V^2$, with $\alpha = 5\,atm/m^6$. The gas is expanded to twice its original volume of $1\,m^3$. How much work is done by the expanding gas in this processView Solution
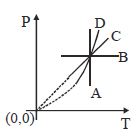
- 3Following figure shows $P-T$ graph for four processes $A, B, C$ and $D$. Select the correct alternativeView Solution
- 4$110\; J$ of heat is added to a gaseous system, whose internal energy change is $40\; J$, then the amount of external work done is ........ $J$View Solution
- 5A carnot engine has the same efficiency between $800 K$ to $500 K$ and $x\, K$ to $600 K.$ The value of $x$ is ...... $K$View Solution
- 6Following figure shows $P-T$ graph for four processes $A, B, C$ and $D$. Select the correct alternativeView Solution
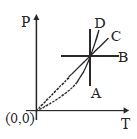
- 7View SolutionThe internal energy of the gas increases In
- 8View SolutionA measure of the degree of disorder of a system is known as
- 9The coefficient of performance of a Carnot refrigerator working between ${30^o}C$ and ${0^o}C$ isView Solution
- 10View SolutionIn adiabatic expansion
