Heat is supplied to a diatomic gas at constant pressure. The ratio of $\Delta Q\,:\,\Delta U\,:\,\Delta W$ is
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A bubble from bottom of lake rises to its surface. Its volume doubles in the process. Assuming isothermal conditions, atmospheric pressure $= 75\, cm$ of $Hg$ and ratio of densities of mercury and water $40/3$. The depth of lake will be ..... $m$View Solution
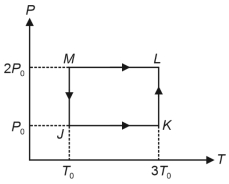
- 2One mole of a monatomic ideal gas undergoes the cyclic process $J \rightarrow K \rightarrow L \rightarrow M \rightarrow J$, as shown in the $P - T$ diagram.View Solution
Match the quantities mentioned in $List-I$ with their values in $List-II$ and choose the correct option. [ $R$ is the gas constant]
$List-I$ $List-II$ ($P$) Work done in the complete cyclic process ($1$) $R T_0-4 \ R T_0 \ln 2$ ($Q$) Change in the internal energy of the gas in the process $JK$ ($2$) $0$ ($R$) Heat given to the gas in the process $KL$ ($3$) $3 \ R T_0$ ($S$) Change in the internal energy of the gas in the process $MJ$ ($4$) $-2 \ R T_0 \ln 2$ ($5$) $-3 \ R T_0 \ln 2$ 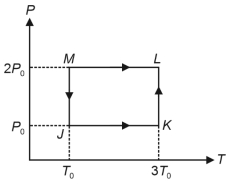
- 3Match List $I$ with List $II$ :View Solution
List $I$ List $II$ $A$ Isothermal Process $I$ Work done by the gas decreases internal energy $B$ Adiabatic Process $II$ No change in internal energy $C$ Isochoric Process $III$ The heat absorbed goes partly to increase internal energy and partly to do work $D$ Isobaric Process $IV$ No work is done on or by the gas Choose the correct answer from the options given below :
- 4View SolutionThe adiabatic Bulk modulus of a diatomic gas at atmospheric pressure is
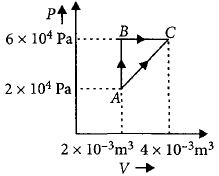
- 5Figure below shows two paths that may be taken by a gas to go from a state $A$ to a state $C.$ In process $AB,$ $400 \,J$ of heat is added to the system and in process $BC,$ $100\, J$ of heat is added to the system. The heat absorbed by the system in the process $AC$ will be ...... $J$View Solution
- 6A closed vessel contains $0.1$ mole of a monoatomic ideal gas at $200\, K$. If $0.05$ mole of the same gas at $400\, K$ is added to it, the final equilibrium temperature (in $K$ ) of the gas in the vessel will be closed toView Solution
- 7View SolutionHeat is not being exchanged in a body. If its internal energy is increased, then
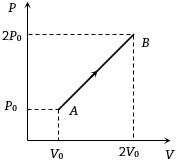
- 8The $P-V$ diagram of $2$ gm of helium gas for a certain process $A \to B$ is shown in the figure. what is the heat given to the gas during the process $A \to B$View Solution
- 9The specific heat capacity of a metal at low temperature $(T)$ is given as $C_p=32\left(\frac{ T }{400}\right)^{3}\;kJ\,k ^{-1}\, kg ^{-1}$. A $100\; g$ vessel of this metal is to be cooled from $20 \;K$ to $4\; K$ by a special refrigerator operating at room temperature $27^\circ c$). The amount of work required to cool the vessel isView Solution
- 10One mole of a monatomic ideal gas undergoes the cyclic process $J \rightarrow K \rightarrow L \rightarrow M \rightarrow J$, as shown in the $P - T$ diagram.View Solution
Match the quantities mentioned in $List-I$ with their values in $List-II$ and choose the correct option. [ $R$ is the gas constant]
$List-I$ $List-II$ ($P$) Work done in the complete cyclic process ($1$) $R T_0-4 \ R T_0 \ln 2$ ($Q$) Change in the internal energy of the gas in the process $JK$ ($2$) $0$ ($R$) Heat given to the gas in the process $KL$ ($3$) $3 \ R T_0$ ($S$) Change in the internal energy of the gas in the process $MJ$ ($4$) $-2 \ R T_0 \ln 2$ ($5$) $-3 \ R T_0 \ln 2$