The pressure and temperature of an ideal gas in a closed vessel are $720\, kPa$ and $40^oC$ respectively. If $\frac{1}{4}^{th}$ of the gas is released from the vessel and the temperature of the remaining gas is raised to $353^oC,$ the final pressure of the gas is ....... $kPa$
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1$40$ calories of heat is needed to raise the temperature of $1\, mole$ of an ideal monoatomic gas from $20°C$ to $30°C$ at a constant pressure. The amount of heat required to raise its temperature over the same interval at a constant volume $(R = 2\,calorie\,mol{e^{ - 1}}{K^{ - 1}})$ is ..... $calorie$View Solution
- 2A container $X$ has volume double that of contianer $Y$ and both are connected by a thin tube. Both contains same ideal gas. The temperature of $X$ is $200\,\,K$ and that of $Y$ is $400\,\,K$. If mass of gas in $X$ is $m$ then in $Y$ it will be:View Solution
- 3View SolutionThe specific heat of a gas in a polytropic process is given by
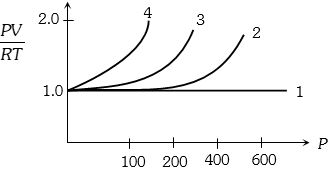
- 4A fix amount of nitrogen gas ($1$ mole) is taken and is subjected to pressure and temperature variation. The experiment is performed at high pressure as well as high temperatures. The results obtained are shown in the figures. The correct variation of $PV/RT$ with $P$ will be exhibited byView Solution
- 5In the two vessels of same volume, atomic hydrogen and helium at pressure $1\, atm$ and $2\, atm$ are filled. If temperature of both the samples is same, then average speed of hydrogen atoms $ < {C_H} > $ will be related to that of helium $ < {C_{He}} > $ asView Solution
- 6The temperature of the hydrogen at which the average speed of its molecules is equal to that of oxygen molecules at a temperature of $31\,^oC,$ is ........ $^oC$View Solution
- 7View SolutionThe vapour of a substance behaves as a gas
- 8A cylinder made of perfectly non conducting material closed at both ends is divided into two equal parts by a heat proof piston. Both parts of the cylinder contain the same masses of a gas at a temperature $t_0 = 27^o$ and pressure $P_0 = 1$ atm. Now if the gas in one of the parts is slowly heated to $t = 57^oC$ while the temperature of first part is maintained at $t_0$ the distance moved by the piston from the middle of the cylinder will be.... $cm$ (length of the cylinder $= 84\,\, cm$)View Solution
- 9If the molecular weight of two gases are $M_1$ and $ M_1$, then at a temperature the ratio of root mean square velocity $v_1$ and $v_2$ will beView Solution
- 10One mole of monatomic gas and three moles of diatomic gas are put together in a container. The molar specific heat (in $JK ^{-1} mol ^{-1}$ ) at constant volume is (Let $R=8 \,JK ^{-1} mol ^{-1}$ )View Solution
