The temperature of the hydrogen at which the average speed of its molecules is equal to that of oxygen molecules at a temperature of $31\,^oC,$ is ........ $^oC$
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
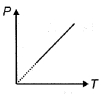
- 1The pressure $P$ of an ideal diatomic gas varies with its absolute temperature $T$ as shown in figure. The molar heat capacity of gas during this process is ........... $R$ [$R$ is gas constant]View Solution
- 2A gas mixture consists of $3\,moles$ of oxygen and $5\,moles$ or argon at temperature $T.$ Considering only translational and rotational modes, the total internal energy of the system isView Solution
- 3An ideal gas $(\gamma = 1.5)$ is expanded adiabatically. How many times has the gas to be expanded to reduce the root mean square velocity of molecules $2.0$ timesView Solution
- 4The total kinetic energy of $1$ mole of oxygen at $27^{\circ} \mathrm{C}$ is :View Solution
[Use universal gas constant $(R)=8.31 \mathrm{~J} / \mathrm{mole} \mathrm{K}$ ]
- 5View SolutionWhich of the following statements about kinetic theory of gases is wrong
- 6Two moles of helium are mixed with $n$ with moles of hydrogen. If $\frac{{{C_P}}}{{{C_V}}}\, = \,\frac{3}{2}$ for the mixture, then the value of $n$ isView Solution
- 7The temperature of a gas is $-50^{\circ}\,C$. To what temperature the gas should be heated so that the rms speed is increased by $3$ times?View Solution
- 8$2\, kg$ of a monoatomic gas is at a pressure of $4\times10^4\, N/m^2$. The density of the gas is $8\, kg/m^3$. What is the order of energy of the gas due to its thermal motion ?View Solution
- 9Hydrogen gas and oxygen gas have volume $1 \,\,cm^3$ each at $N.T.P$.View Solution
- 10An ideal gas is initially at temperature $T$ and volume $V.$ Its volume is increased by $\Delta V$ due to an increase in temperature $\Delta T,$ pressure remaining constant. The quantity $\delta = \Delta V/(V\Delta T)$ varies with temperature asView Solution
