$3\,moles$ of an ideal gas at a temperature of $27^{\circ}\,C$ are mixed with $2\,moles$ of an ideal gas at a temperature $227^{\circ}\,C$, determine the equilibrium temperature (${}^o C$) of the mixture, assuming no loss of energy.
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Assertion $(A):$ The total translational kinetic energy of all the molecules of a given mass of an ideal gas is $1.5$ times the product of its pressure and volume.View Solution
Reason $(R):$ The molecules of gas collide with each other and the velocities of the molecules change due to the collision.
- 2If the r.m.s. speed of chlorine molecule is $490\,m / s$ at $27^{\circ}\,C$, the r.m.s. speed of argon molecules at the same temperature will be $......\,m/s$ (Atomic mass of argon $=39.9\,u$, molecular mass of chlorine $=70.9\,u )$View Solution
- 3The pressure and volume of saturated water vapour are $P$ and $V$ respectively. It is compressed isothermally thereby volume becomes $V/2,$ the final pressure will beView Solution
- 4Three perfect gases at absolute temperatures $T_1 , T_2$ and $T_3$ are mixed. The masses of molecules are $m_1 , m_2$ and $m_3$ and the number of molecules are $n_1, n_2$ and $n_3$ respectively. Assuming no loss of energy, the final temperature of the mixture isView Solution
- 5The root mean square speed of smoke particles of mass $5 \times 10^{-17}\,kg$ in their Brownian motion in air at NTP is approximately $.......\,mm\,s ^{-1}$ [Given $k =1.38 \times 10^{-23}\,J\,K ^{-1}$ ]View Solution
- 6The temperature of a gas is $-78^{\circ} \mathrm{C}$ and the average translational kinetic energy of its molecules is $\mathrm{K}$. The temperature at which the average translational kinetic energy of the molecules of the same gas becomes $2 \mathrm{~K}$ is :View Solution
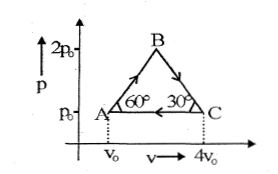
- 7Four moles of an ideal monoatomic gas undergoes a cyclic process $ABCA$ as shown in figure find ratio of temperature of $A$ and $B$ .View Solution
- 8View SolutionThe molecules of an ideal gas at a certain temperature have
- 9The ratio of specific heats $(\gamma )$ of a ideal gas is given byView Solution
- 10View SolutionThe r.m.s. speed of gas molecules is given by
