The ratio of average translational kinetic energy to rotational kinetic energy of a diatomic molecule at temperature $T$ is
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1For a gas the difference between the two specific heats is $4150\, J/kg\, K.$ What is the specific heats at constant volume of gas if the ratio of specific heat is $1.4$View Solution
- 2View SolutionAt constant volume, temperature is increased. Then
- 3Two gases occupy two containers $A$ and $B$ the gas in $A$, of volume $0.10\,m ^3$, exerts a pressure of $1.40\,MPa$ and that in $B$ of volume $0.15 m ^3$ exerts a pressure $0.7\,MPa$. The two containers are united by a tube of negligible volume and the gases are allowed to intermingle. Then if the temperature remains constant, the final pressure in the container will be (in MPa)View Solution
- 4A cubical box with porous walls containing an equal number of ${O_2}$ and $H_2$ molecules is placed in a large evacuated chamber. The entire system is maintained at constant temperature $T.$ The ratio of ${v_{rms}}$ of ${O_2}$ molecules to that of the ${v_{rms}}$ of $H_2$ molecules, found in the chamber outside the box after a short interval isView Solution
- 5At a given temperature the $r.m.s.$ velocity of molecules of the gas isView Solution
- 6The pressure and temperature of an ideal gas in a closed vessel are $720\, kPa$ and $40^oC$ respectively. If $\frac{1}{4}^{th}$ of the gas is released from the vessel and the temperature of the remaining gas is raised to $353^oC,$ the final pressure of the gas is ....... $kPa$View Solution
- 7In a dilute gas at pressure $P$ and temperature $T$, the mean time between successive collisions of a molecule varies with $T$ asView Solution
- 8View SolutionA container with rigid walls is covered with perfectly insulating material. The container is divided into two parts by a partition. One part contains a gas while the other is fully evacuated (vacuum). The partition is suddenly removed. The gas rushes to fill the entire volume and comes to equilibrium after a little time. If the gas is not ideal, then
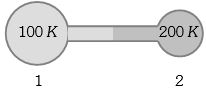
- 9Figure shows two flasks connected to each other. The volume of the flask $1$ is twice that of flask $2.$ The system is filled with an ideal gas at temperature $100\, K$ and $200 \,K $ respectively. If the mass of the gas in $1$ be $m$ then what is the mass of the gas in flask $2$View Solution
- 10An engineer is given a fixed volume $V_m$ of metal with which to construct a spherical pressure vessel. Interestingly, assuming the vessel has thin walls and is always pressurized to near its bursting point, the amount of gas the vessel can contain, $n$ (measured in moles), does not depend on the radius $r$ of the vessel : instead it depends only on $V_m$ (measured in $m^3$) the temperature $T$ (mensured in $K$). the ideal gas constant $R$ (measured in $J/(K\ mol$ )), and the tensile strength of the metal $\sigma $ (measured in $N/m^2$ ) . Which of the following gives $n$ in terms of these parameters?View Solution
