The relation between two specific heats of a gas is
Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
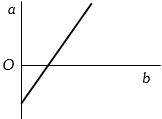
- 1View SolutionThe expansion of unit mass of a perfect gas at constant pressure is shown in the diagram. Here
- 2View SolutionEvery gas (real gas) behaves as an ideal gas
- 3A diatomic gas of molecular mass $40 \,g / mol$ is filled in a rigid container at temperature $30^{\circ} C$. It is moving with velocity $200 \,m / s$. If it is suddenly stopped, the rise in the temperature of the gas is .........View Solution
- 4Five moles of helium are mixed with two moles of hydrogen to form a mixture. Take molar mass of helium $M_1=4\ g$ and that of hydrogen $M_2=2\ g$ If the internal energy of He sample of $100\,\,J$ and that of the hydrogen sample is $200\,\,J$, then the internal energy of the mixture is ..... $J$View Solution
- 5The equation of a certain gas can be written as: ${\left( {\frac{{{T^7}}}{{{P^2}}}} \right)^{1/5}} = $ constant. The specific heat at constant volume of this gas is in $\left( {in\frac{J}{{mol\,K}}} \right)$View Solution
- 6View SolutionAn ideal gas is filled in a closed container and container is moving with uniform acceleration in horizontal direction. Neglect gravity. Pressure inside the container is ...............
- 7The value of critical temperature in terms of Vander Waal’s constant $a$ and $b$ isView Solution
- 8A vessel contains a mixture of one mole of oxygen and two moles of nitrogen at $300 K$. The ratio of the average rotational kinetic energy per ${O_2}$ molecule to that per ${N_2}$ molecule isView Solution
- 9What is the mass of $2$ litres of nitrogen at $22.4$ atmospheric pressure and $273\,K$View Solution
- 10A box contains n molecules of a gas. How will the pressure of the gas be effected, if the number of molecules is made $2n$View Solution
