What is the mass of $2$ litres of nitrogen at $22.4$ atmospheric pressure and $273\,K$
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1The equation of state of a gas is given by $\left( {P + \frac{{a{T^2}}}{V}} \right)\,{V^c} = (RT + b)$, where $a, b, c$ and $R$ are constants. The isotherms can be represented by $P = A{V^m} - B{V^n}$, where $A$ and $B$ depend only on temperature thenView Solution
- 2An $HCl$ molecule has rotational, translational and vibrational motions. If the $rms$ velocity of $HCl$ molecules in its gaseous phase is $\bar v ,\,m$ is its mass and $k_B$ is Boltzmann constant, then its temperature will beView Solution
- 3The ratio of mean kinetic energy of hydrogen and nitrogen at temperature $300 \,K$ and $450\, K$ respectively isView Solution
- 4A cylinder made of perfectly non conducting material closed at both ends is divided into two equal parts by a heat proof piston. Both parts of the cylinder contain the same masses of a gas at a temperature $t_0 = 27^o$ and pressure $P_0 = 1$ atm. Now if the gas in one of the parts is slowly heated to $t = 57^oC$ while the temperature of first part is maintained at $t_0$ the distance moved by the piston from the middle of the cylinder will be.... $cm$ (length of the cylinder $= 84\,\, cm$)View Solution
- 5A mixture of ideal gases has $2$ moles of He, $4$ moles of oxygen and $1$ mole of ozone at absolute temperature $T$. The internal energy of mixture is ............. $RT$View Solution
- 6View SolutionThe specific heat of an ideal gas is
- 7Molecules of an ideal gas are known to have three translational degrees of freedom and two rotational degrees of freedom. The gas is maintained at a temperature of $T$. The total internal energy, $U$ of a mole of this gas, and the value of $\gamma\left(=\frac{ C _{ P }}{ C _{ v }}\right)$ given, respectively, byView Solution
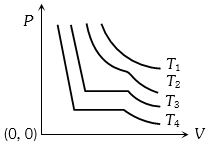
- 8The change in volume $V$ with respect to an increase in pressure $P$ has been shown in the figure for a non-ideal gas at four different temperatures ${T_1},\,{T_2},\,{T_3}$ and ${T_4}$. The critical temperature of the gas isView Solution
- 9A thermally insulated vessel contains an ideal gas of molecular mass $M$ and ratio of specific heats $\gamma$. It is moving with speed $v$ and it's suddenly brought to rest. Assuming no heat is lost to the surroundings, its temperature increases byView Solution
- 10On giving equal amount of heat at constant volume to $1\, mol$ of a monoatomic and a diatomic gas the rise in temperature $(\Delta T)$ is more forView Solution
