Helium at ${27^o}C$ has a volume of $8$ litres. It is suddenly compressed to a volume of $1$ litre. The temperature of the gas will be ....... $^oC$ $[\gamma = 5/3]$
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1During the melting of a slab of ice at $273\, K$ at atmospheric pressureView Solution
- 2Unit mass of a liquid with volume ${V_1}$ is completely changed into a gas of volume ${V_2}$ at a constant external pressure $P$ and temperature $T.$ If the latent heat of evaporation for the given mass is $L,$ then the increase in the internal energy of the system isView Solution
- 3Diatomic gas is used in carnot heat engine. If efficiency of given carnot heat engine is $80\%$ , then find the ratio of initial volume to final volume of gas during adiabatic expansionView Solution
- 4View SolutionIn an isothermal process the volume of an ideal gas is halved. One can say that
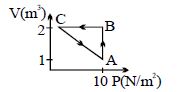
- 5An ideal gas is taken through the cycle $A \to B \to C \to A$ , as shown in the figure. If the net heat supplied to the gas in the cycle is $5\ J$, the work done by the gas in the process $C \to A$ is .... $J$View Solution
- 6View SolutionWhich of the following parameters does not characterize the thermodynamic state of matter
- 7Let $\eta_{1}$ is the efficiency of an engine at $T _{1}=447^{\circ}\,C$ and $T _{2}=147^{\circ}\,C$ while $\eta_{2}$ is the efficiency at $T _{1}=947^{\circ}\,C$ and $T _{2}=47^{\circ}\,C$. The ratio $\frac{\eta_{1}}{\eta_{2}}$ will be.View Solution
- 8A diatomic gas $(\gamma=1.4)$ does $200 \mathrm{~J}$ of work when it is expanded isobarically. The heat given to the gas in the process is:View Solution
- 9The internal energy change in a system that has absorbed $2 \;k cal$ of heat and done $500 \;J $ of work is ...... $J$View Solution
- 10A Carnot engine, whose efficiency is $40\%$, takes in heat from a source maintained at a temperature of $500\ K$. It is desired to have an engine of efficiency $60\%$. Then, the intake temperature for the same exhaust (sink) temperature must be ....... $K$View Solution
