The specific heat of hydrogen gas at constant pressure is ${C_P} = 3.4 \times {10^3}cal/kg{\,^o}C$ and at constant volume is ${C_V} = 2.4 \times {10^3}cal/kg{\,^o}C.$If one kilogram hydrogen gas is heated from ${10^o}C$ to ${20^o}C$ at constant pressure, the external work done on the gas to maintain it at constant pressure is
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A system performs work $\Delta W$ when an amount of heat is$\Delta Q$ added to the system, the corresponding change in the internal energy is $\Delta U$. A unique function of the initial and final states (irrespective of the mode of change) isView Solution
- 2A thermodynamic process is shown in the figure. The pressures and volumes corresponding to some points in the figure are :View Solution
${P_A} = 3 \times {10^4}Pa,\;{P_B} = 8 \times {10^4}Pa$ and ${V_A} = 2 \times {10^{ - 3}}{m^3},\;{V_D} = 5 \times {10^{ - 3}}{m^3}$
In process $AB$, $600 J$ of heat is added to the system and in process $BC, 200 J $ of heat is added to the system. The change in internal energy of the system in process $ AC$ would be ...... $J$
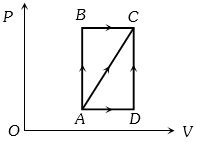
- 3In a thermodynamic process two moles of a monatomic ideal gas obeys $P \propto V^{-2}$. If temperature of the gas increases from $300 \,K$ to $400 \,K$, then find work done by the gas .........$R$ (where $R=$ universal gas constant).View Solution
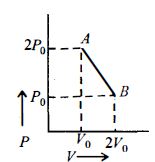
- 4$n\, moles$ of an ideal gas undergo a process $A \to B$ as shown in the figure. Maximum temperature of the gas during the process isView Solution
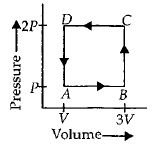
- 5A thermodynamic system is taken through the cycle $ABCD$ as shown in figure. Heat rejected by the gas during the cycle isView Solution
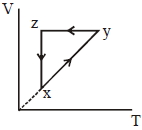
- 6A thermodynamic cycle $xyzx$ is shown on a $V-T$ diagram.View Solution
The $P-V$ diagram that best describes this cycle is
(Diagrams are schematic and not to scale)
- 7Two Carnot engines $A$ and $B$ are operated in succession. The first one, $A$ receives heat from a source at ${T_1} = 800K$ and rejects to sink at ${T_2}K.$. The second engine $B$ receives heat rejected by the first engine and rejects to another sink at ${T_3} = 300K.$ If the work outputs of two engines are equal, then the value of ${T_2}$ is .... $K$View Solution
- 8Even Carnot engine cannot give $100\%$ efficiency because we cannotView Solution
- 9A fixed amount of a gas undergoes a thermodynamic process as shown such that heat interaction along path $B \to C \to A$ is equal to the work done by the gas along path $A \to B \to C$. Then process $A \to B$ is :-View Solution
- 10One mole of an ideal gas is taken through an adiabatic process where the temperature rises from $27^{\circ} {C}$ to $37^{\circ} {C}$. If the ideal gas is composed of polyatomic molecule that has $4$ vibrational modes which of the following is true?View Solution
