The specific heat of isothermal process is
Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
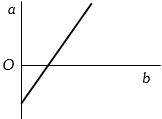
- 1View SolutionThe expansion of unit mass of a perfect gas at constant pressure is shown in the diagram. Here
- 2View SolutionWhich of the following statements about kinetic theory of gases is wrong
- 3A diatomic gas of molecular mass $40 \,g / mol$ is filled in a rigid container at temperature $30^{\circ} C$. It is moving with velocity $200 \,m / s$. If it is suddenly stopped, the rise in the temperature of the gas is .........View Solution
- 4Match List$-I$ with List$-II:$View Solution
List$-I$ List$-II$ $(A)$ $3$ Translational degrees of freedom $(I)$ Monoatomic gases $(B)$ $3$ Translational,$2$ rotational degrees of freedoms $(II)$ Polyatomic gases $(C)$ $3$ Translational,$2$ rotational and $1$ vibrational degrees of freedom $(III)$ Rigid diatomic gases $(D)$ $3$ Translational,$3$ rotational and more than one vibrational degrees of freedom $(IV)$ Nonrigid diatomic gases Choose the correct answer from the options given below:
- 5An insulated container contains $4\, moles$ of an ideal diatomic gas at temperature $T$. Heat $Q$ is supplied to this gas, due to which $2\, moles$ of gas are dissociated into atoms but temperature of the gas remains constant. Then:View Solution
- 6View SolutionIf the mean free path of atoms is doubled then the pressure of gas will become
- 7What is/are the same for $O_2$ and $NH_3$ in gaseous stateView Solution
- 8One mole of an ideal gas at $STP$ is heated in an insulated closed container until the average speed of its molecules is doubled. Its pressure would therefore increase by factor.View Solution
- 9Five moles of helium are mixed with two moles of hydrogen to form a mixture. Take molar mass of helium $M_1=4\ g$ and that of hydrogen $M_2=2\ g$ The equivalent value of $\gamma$ isView Solution
- 10Hydrogen gas and oxygen gas have volume $1 \,\,cm^3$ each at $N.T.P$.View Solution
