What is/are the same for $O_2$ and $NH_3$ in gaseous state
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Vessel $A$ is filled with hydrogen while vessel $B,$ whose volume is twice that of $A,$ is filled with the same mass of oxygen at the same temperature. The ratio of the mean kinetic energies of hydrogen and oxygen isView Solution
- 2A cylinder of fixed capacity of $44.8 \,litres$ contains helium gas at standard temperature and pressure. The amount of heat needed to raise the temperature of gas in the cylinder by $20.0^{\circ} C$ will be .............. $J$(Given gas constant $R =8.3 \,JK ^{-1}- moI ^{-1}$ )View Solution
- 3Equation of gas in terms of pressure $(P),$ absolute temperature $(T)$ and density $(d)$ isView Solution
- 4View SolutionOn absolute temperature, the kinetic energy of the molecules
- 5Hydrogen gas and oxygen gas have volume $1 \,\,cm^3$ each at $N.T.P$.View Solution
- 6The value closest to the thermal velocity of a Helium atom at room temperature $(300\,K)$in $ms^{-1}$ is $[k_B\, = 1 .4\times10^{-23}\,J/K;\, m_{He}\, = 7\times10^{-27}\,kg]$View Solution
- 7What will be the average value of energy along one degree of freedom for an ideal gas in thermal equilibrium at a temperature $T \,?\left( k _{ B }\right.$ is Boltzmann constant)View Solution
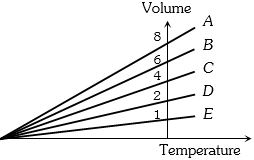
- 8The expansion of an ideal gas of mass $m$ at a constant pressure $P$ is given by the straight line $D$. Then the expansion of the same ideal gas of mass $2m$ at a pressure $P/ 2 $ is given by the straight lineView Solution
- 9The gas in vessel is subjected to a pressure of $20$ atmosphere at a temperature $27°C.$ The pressure of the gas in a vessel after one half of the gas is released from the vessel and the temperature of the remainder is raised by $50°C$ is ....... $atm$View Solution
- 10View SolutionYou have two containers of equal volume. One is full of helium gas. The other holds an equal mass of nitrogen gas. Both gases have the same pressure. How does the temperature of the helium compare to the temperature of the nitrogen ?
