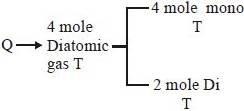
An insulated container contains $4\, moles$ of an ideal diatomic gas at temperature $T$. Heat $Q$ is supplied to this gas, due to which $2\, moles$ of gas are dissociated into atoms but temperature of the gas remains constant. Then:
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1The ratio of two specific heats $\frac{{{C_P}}}{{{C_V}}}$ of $CO$ isView Solution
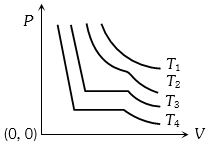
- 2The change in volume $V$ with respect to an increase in pressure $P$ has been shown in the figure for a non-ideal gas at four different temperatures ${T_1},\,{T_2},\,{T_3}$ and ${T_4}$. The critical temperature of the gas isView Solution
- 3At a temperature $T K,$ the pressure of $4.0\,\,g$ argon in a bulb is $p$. The bulb is put in a bath having temperature higher by $50\,\,K$ than the first one. $0.8\,\,g$ of argon gas had to be removed to maintained original pressure. The temperature $T$ is equal to ..... $K$View Solution
- 4At room temperature, the $r.m.s.$ speed of the molecules of certain diatomic gas is found to be $1920\, m/s$. The gas isView Solution
- 5View SolutionThe kinetic energy per gm mol for a diatomic gas at room temperature is
- 6View SolutionThe parameter that remains the same for molecules of all gases at a given temperature is :
- 7At what temperature is the root mean square velocity of gaseous hydrogen molecules is equal to that of oxygen molecules at $47°C$ ..... $K$View Solution
- 8$c_P$ and $c_V$ are specific heats at constant pressure and constant volume respectively. It is observed thatView Solution
$c_P - c_V = a$ for hydrogen gas
$c_P - c_V = b$ for nitrogen gas
The correct relation between $a$ and $b$ is
- 9A flask contains argon and oxygen in the ratio of $3: 2$ in mass and the mixture is kept at $27^{\circ} C$. The ratio of their average kinetic energy per molecule respectively will be ...........View Solution
- 10The temperature, at which the root mean square velocity of hydrogen molecules equals their escape velocity from the earth is closest to : [Boltzmans Constant $k_B = 1.38\times10^{-23}\, J / K$ Avogadro number $N_A = 6.02\times10^{26}\, / kg$ Radius of Earth $: 6.4\times10^6\, m$ Gravitation acceleration on Earth $= 10\, ms^{-2}$]View Solution