The temperature at which the $r.m.s.$ speed of hydrogen molecules is equal to escape velocity on earth surface, will be ...... $K$
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1If $C_p$ and $C_v$ denote the specific heats of nitrogen per unit mass at constant pressure and constant volume respectively, thenView Solution
- 2A $15\,g$ mass of nitrogen gas is enclosed in a vessel at a temperature $27\,^oC.$ Amount of heat transferred to the gas, so that rms velocity of molecules is doubled, is about ...... $kJ$ [Take $R = 8.3\,J/K\,mole$ ]View Solution
- 3A vertical closed cylinder is separated into two parts by a frictionless piston of mass $m$ and of negligible thickness. The piston is free to move along the length of the cylinder .The length of the cylinder above the piston is $l_1,$ and that below the piston is $l_2,$ such that $l_1 > l_2.$ Each part of the cylinder contains $n$ moles of an ideal gas at equal temperature $T.$ If the piston is stationary, its mass, $m,$ will be given by: ( $R$ is universal gas constant and $g$ is the acceleration due to gravity)View Solution
- 4A gas mixture consists of $2$ moles of $O_2$ and $4$ moles of $Ar$ at temperature $T$. Neglecting all vibrational modes, the total internal energy of the system isView Solution
- 5An air bubble doubles its radius on raising from the bottom of water reservoir to be the surface of water in it. If the atmospheric pressure is equal to $10\, m$ of water, the height of water in the reservoir is ..... $m$View Solution
- 6View SolutionThe root mean square speed of the molecules of a gas is
- 7Under an adiabatic process, the volume of an ideal gas gets doubled. Consequently the mean collision time between the gas molecule changes from $\tau_{1}$ to $\tau_{2} .$ If $\frac{C_{p}}{C_{v}}=\gamma$ for this gas then a good estimate for $\frac{\tau_{2}}{\tau_{1}}$ is given by :View Solution
- 8On any planet, the presence of atmosphere implies (${C_{rms}}$= root mean square velocity of molecules and ${V_e}$= escape velocity)View Solution
- 9For a given gas at $1\,atm$ pressure, $rms$ speed of the molecules is $200\,m/s$ at $127\,^oC.$ At $2\,atm$ pressure and at $227\,^oC,$ the $rms$ speed of the molecules will beView Solution
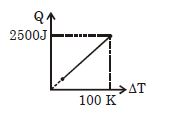
- 10One mole of a gas mixture is heated under constant pressure, and heat supplied $Q$ is plotted against temperature difference acquired. Find the approximate value of $\gamma $ for mixtureView Solution