The respective speeds of five molecules are $2, 1.5, 1.6, 1.6$ and $1.2 \,km/sec.$ The most probable speed in $km/sec$ will be
Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1The temperature of argon, kept in a vessel, is raised by $1^\circ C$ at a constant volume. The total heat supplied to the gas is a combination of translational and rotational energies. Their respective shares areView Solution
- 2In Maxwell's speed distribution curve, for $N_2$ gas, the average of $\mid$ relative velocity (in $m/s$) $\mid$ between two molecules at $300 \,K$ will beView Solution
- 3View SolutionAt absolute zero temperature, pressure of a gas will be
- 4When the temperature of a gas is raised from $30^o C$ to $90^o C$ , the percentage increase in the $r.m.s.$ velocity of the molecules will beView Solution
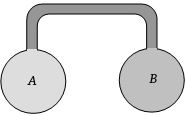
- 5Two identical adiabatic vessels are filled with oxygen at pressure $P_1$ and $P_2 (P_1 > P_2).$ The vessels are interconnected with each other by a nonconducting pipe. If $U_{01}$ and $U_{02}$ denote initial internal energy of oxygen in first and second vessel respectively and $U_{f_1}$ and $U_{f_2}$ denote final internal energy values, than :View Solution
- 6View SolutionThe molecules of air in the room that you are sitting are all experiencing the force of gravity tending to bring them down. The molecules are also frequently and randomly undergoing collisions, which tend to oppose the effect of fall under gravity. The density of air is nearly uniform throughout the room because
- 7If a given mass of gas occupies a volume of $10\, cc$ at $1$ atmospheric pressure and temperature of $100°C(373.15\, K).$ What will be its volume at 4 atmospheric pressure; the temperature being the same .... $cc$View Solution
- 8When one mole of monatomic gas is mixed with one mole of a diatomic gas, then the equivalent value of $\gamma$ for the mixture will be (vibration mode neglected)View Solution
- 9Two spherical vessel of equal volume, are connected by a n arrow tube. The apparatus contains an ideal gas at one atmosphere and $300K$. Now if one vessel is immersed in a bath of constant temperature $600K$ and the other in a bath of constant temperature $300K$. Then the common pressure will be ...... $atm$View Solution
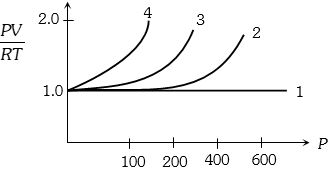
- 10A fix amount of nitrogen gas ($1$ mole) is taken and is subjected to pressure and temperature variation. The experiment is performed at high pressure as well as high temperatures. The results obtained are shown in the figures. The correct variation of $PV/RT$ with $P$ will be exhibited byView Solution