$0.25=1-\frac{\mathrm{T}_{2}}{\mathrm{T}_{1}} \Rightarrow \frac{1}{4}=1-\frac{\mathrm{T}_{2}}{\mathrm{T}_{1}}$
$\frac{\mathrm{T}_{2}}{\mathrm{T}_{1}}=1-\frac{1}{4}=\frac{3}{4}$ $ . .(\mathrm{i})$
According to question.
$\eta_{2}=2 \eta_{1},$ and $\mathrm{T}_{2}=\mathrm{T}_{2}-58^{\circ} \mathrm{C}$
$\therefore \quad 2 \times \frac{1}{4}=1-\frac{\left(\mathrm{T}_{2}-58^{\circ} \mathrm{C}\right)}{\mathrm{T}_{1}}$
$\Rightarrow 1-\frac{1}{2}=\frac{\mathrm{T}_{2}-58^{\circ} \mathrm{C}}{\mathrm{T}_{1}}$
$\frac{1}{2}=\frac{T_{2}}{T_{1}}-\frac{58^{\circ}}{T_{1}} \Rightarrow \frac{3}{4}-\frac{1}{2}=\frac{58}{T_{1}}$
$\Rightarrow \mathrm{T}_{1}=232^{\circ} \mathrm{C}$
Download our appand get started for free
Similar Questions
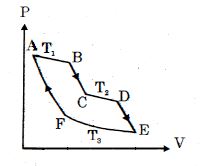
- 1One mole of ideal gas taken through a cycle process with alternate isothermal and adiabatic curves. In $P-V$ diagram $AB, CD, EF$ are isothermal curves at the absolute temperature $T_1, T_2$ and $T_3$ respectively and $BC, DE$ and $FA$ are adiabatic curves respectively. If $\frac{{{V_B}}}{{{V_A}}} = 2,\,\frac{{{V_D}}}{{{V_C}}} = 2$ then for cycle is shown in figure four statements are being made given below. (Figure is not drawn on scale)View Solution
Statement $1$ : Ratio of volumes $\frac{{{V_E}}}{{{V_F}}} = 4$
Statement $2$ : Magnitude of work done in isothermal compression $EF$ is $2RT_3\ ln\ (2)$
Statement $3$ : Ratio of heat supplied to gas in the process $AB$ to heat rejected by gas in process $EF$ is $\frac{{{T_1}}}{{2{T_3}}}$
Statement $4$ : Net work done by gas in the cycle $ABCDEFA$ is $(T_1 + T_2 - 2T_3) R\ ln\ (2)$
Find the number of correct statement $(s)$ given for the cyclic process followed by gas
- 2A diatomic gas undergoes a process represented by $PV ^{1.3}=$ constant. Choose the incorrect statementView Solution
- 3View SolutionWhich statement is incorrect?
- 4View SolutionWhich of the following is a slow process
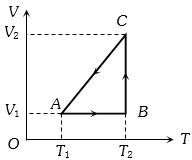
- 5A cyclic process for $1\, mole$ of an ideal gas is shown in figure in the $V-T,$ diagram. The work done in $AB, BC$ and $CA$ respectivelyView Solution
- 6View SolutionAn adiabatic process occurs at constant
- 7$Assertion :$ In an isolated system the entropy increases.View Solution
$Reason :$ The processes in an isolated system are adiabatic. - 8A sample of gas with $\gamma=1.5$ is taken through an adiabatic process in which the volume is compressed from $1200\, {cm}^{3}$ to $300\, {cm}^{3}$. If the initial pressure is $200\, {kPa}$. The absolute value of the workdone by the gas in the process $= \,..... J.$View Solution
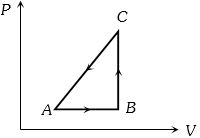
- 9The $P-V$ diagram of a system undergoing thermodynamic transformation is shown in figure. The work done on the system in going from $A → B → C$ is $50 J$ and $ 20\, cal$ heat is given to the system. The change in internal energy between $A$ and $C$ is ...... $J$View Solution
- 10Given that $1\,g$ of water in liquid phase has volume $1\,cm^3$ and in vapour phase $1671\, cm^3$ at atmospheric pressure and the latent heat of vaporization of water is $2256\,J/g;$ the change in the internal energy in joules for $1\,g$ of water at $373\,K$ when it changes from liquid phase to vapour phase at the same temperature is ....... $J$View Solution
