The volume $( V)$ of a monatomic gas varies with its temperature $(T)$ , as shown in the graph. The ratio of work done by the gas , to the heat absorbed by it, when it undergoes a change from state $A$ to state $B$ , is
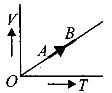

NEET 2018, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionHeat is not being exchanged in a body. If its internal energy is increased, then
- 2$540$ calories of heat convert $1 $ cubic centimeter of water at ${100^o}C$ into $1671 $ cubic centimeter of steam at ${100^o}C$ at a pressure of one atmosphere. Then the work done against the atmospheric pressure is nearly ...... $cal$View Solution
- 3A monatornic gas at a pressure $P,$ having a volume $V$ expands isothermally to a volume $2\, V$ and then adiabatically to a volume $16\, V.$ The final pressure of the gas is $(\,Take \,\gamma = 5/3)$View Solution
- 4A gas mixture consists of $8$ moles of argon and $6$ moles of oxygen at temperature $T$. Neglecting all vibrational modes, the total internal energy of the system isView Solution
- 5A given ideal gas with $\gamma = \frac{{{C_p}}}{{{C_v}}} = 1.5$ at a temperature $T$. If the gas is compressed adiabatically to one-fourth of its initial volume, the final temperature will be ..... $T$View Solution
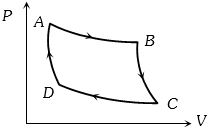
- 6The $P-V$ graph of an ideal gas cycle is shown here as below. The adiabatic process is described byView Solution
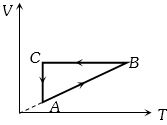
- 7A cyclic process $ABCA$ is shown in the $V-T $ diagram. Process on the $P-V$ diagram isView Solution
- 8A system is given $300$ calories of heat and it does $600$ joules of work. How much does the internal energy of the system change in this process ..... $J$. ($J = 4.18$ joules/cal)View Solution
- 9An ideal system can be brought from state $A$ to $B$ through four paths as shown in the figure. The energy given to the system is minimum inView Solution
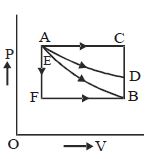
- 10View SolutionA perfect gas contained in a cylinder is kept in vacuum. If the cylinder suddenly bursts, then the temperature of the gas
