$\Rightarrow \mathrm{U}=\mathrm{n}_1 \mathrm{C}_{\mathrm{V}_1} \mathrm{~T}+\mathrm{n}_2 \mathrm{C}_{\mathrm{V}_2} \mathrm{~T}$
$\Rightarrow 8 \times \frac{3 \mathrm{R}}{2} \times \mathrm{T}+6 \times \frac{5 \mathrm{R}}{2} \times \mathrm{T}$
$=27 \mathrm{RT}$
Download our appand get started for free
Similar Questions
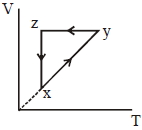
- 1A thermodynamic cycle $xyzx$ is shown on a $V-T$ diagram.View Solution
The $P-V$ diagram that best describes this cycle is
(Diagrams are schematic and not to scale)
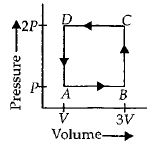
- 2A thermodynamic system is taken through the cycle $ABCD$ as shown in figure. Heat rejected by the gas during the cycle isView Solution
- 3View SolutionIn adiabatic expansion
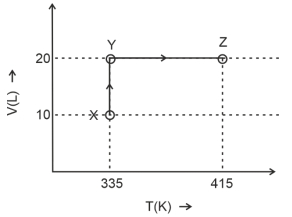
- 4Consider the following volume-temperature $( V - T )$ diagram for the expansion of $5$ moles of an ideal monoatomic gas.View Solution
Considering only $P-V$ work is involved, the total change in enthalpy (in Joule) for the transformation of state in the sequence $X \rightarrow Y \rightarrow Z$ is $\qquad$
[Use the given data: Molar heat capacity of the gas for the given temperature range, $C _{ v , m }=12 J K ^{-1} mol ^{-1}$ and gas constant, $R =8.3 J K ^{-1} mol ^{-1}$ ]
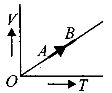
- 5The volume $( V)$ of a monatomic gas varies with its temperature $(T)$ , as shown in the graph. The ratio of work done by the gas , to the heat absorbed by it, when it undergoes a change from state $A$ to state $B$ , isView Solution
- 6Two moles of helium gas are taken over the cycle $ABCDA$, as shown in the $P-T$ diagram.The work done on the gas in taking it from $D$ to $A$ isView Solution

- 7Consider two containers $A$ and $B$ containing monoatomic gases at the same Pressure $(P)$, Volume $(V)$ and Temperature $(T)$. The gas in $A$ is compressed isothermally to $\frac{1}{8}$ of its original volume while the gas $B$ is compressed adiabatically to $\frac{1}{8}$ of its original volume. The ratio of final pressure of gas in $B$ to that of gas in $A$ is ...........View Solution
- 8View SolutionFor free expansion of the gas which of the following is true
- 9Carnot cycle (reversible) of a gas represented by a Pressure-Volume curve is shown in the diagramConsider the following statementsView Solution
$I.$ Area $ABCD =$ Work done on the gas
$II.$ Area $ABCD =$ Net heat absorbed
$III.$ Change in the internal energy in cycle $= 0$
Which of these are correct
- 10In an Isothermal change, the change in pressure and volume of a gas can be represented for three different temperature; $T _3 > T _2 > T _1$ as :View Solution
