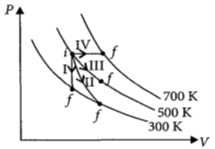
Thermodynamic processes are indicated in the following diagram.
Match the following
$\begin{array}{|l|l|} \hline Column\,\,-\,\,1 & Column\,\,-\,\,2 \\ \hline P\,:\,Process\,\,-\,\,I & \,\,A\,\,:\,\,Adiabatic \\ \hline Q\,:\,Process\,\,-\,\,II & \,\,B\,\,:\,\,Isobaric \\ \hline R\,:\,Process\,\,-\,\,III & \,\,C\,\,:\,\,Isochoric \\ \hline S\,:\,Process\,\,-\,\,IV & \,\,D\,\,:\,\,Isothermal \\ \hline \end{array}$
NEET 2017, Medium
In process $I,$ volume is constant
$\therefore \,\,\,\,process\,I \to Isochoric;\,P \to C$
As slope of curve $II$ is more than the slope of curve $III$.
$process\,II \to Adiabatic\,and\,processIII \to Isothermal$
$\therefore \,\,\,Q \to A,R \to D$
In process $IV$, pressure is constant
Process $IV \to Isobaric;\,S \to B$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A closed vessel contains $0.1$ mole of a monoatomic ideal gas at $200\, K$. If $0.05$ mole of the same gas at $400\, K$ is added to it, the final equilibrium temperature (in $K$ ) of the gas in the vessel will be closed toView Solution
- 2View SolutionWork done on or by a gas, in general depends upon the
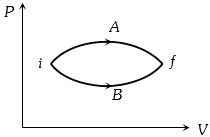
- 3In the figure given two processes $A$ and $B$ are shown by which a thermo-dynamical system goes from initial to final state $F.$ If $\Delta {Q_A}$ and $\Delta {Q_B}$ are respectively the heats supplied to the systems thenView Solution
- 4A Carnot engine working between $300\,K$ and $600\,K$ has work output of $800\, J$ per cycle. What is amount of heat energy supplied to the engine from source per cycle...... $J/cycle$View Solution
- 5An ideal gas has volume ${V_0}$ at ${27^o}C.$ It is heated at constant pressure so that its volume becomes $2{V_0}.$ The final temperature isView Solution
- 6If ${C_V} = 4.96cal/mole\, K$, then increase in internalenergy when temperature of $2$ moles of this gas is increased from $340 K$ to $342 K$ ....... $cal$View Solution
- 7A cylinder of mass $1\,kg$ is given heat of $20000\, J$ at atmospheric pressure. If initially temperature of cylinder is $20\,^oC$, then work done by the cylinder will be .......$J$ (Given that Specific heat of cylinder $= 400 \,J\, kg^{-1}$, Coefficient of volume expansion $= 9 \times {10^{-5}}\,^o C^{-1}$, Atmospheric pressure $= 10^5 \,N/m^2$ and density of cylinder $9000\,kg/m^3$)View Solution
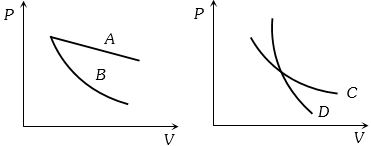
- 8In the following figure, four curves $A, B, C$ and $D$ are shown. The curves areView Solution
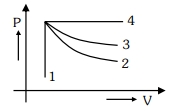
- 9An ideal gas undergoes four different processes from the same initial state as shown in the figure below. Those processes are adiabatic, isothermal, isobaric and isochoric. The curve which represents the adiabatic process among $1,2,3$ and $4$ isView Solution
- 10$Assertion :$ The heat supplied to a system is always equal to the increase in its internal energy.View Solution
$Reason :$ When a system changes from one thermal equilibrium to another, some heat is absorbed by it.
