To an ideal triatomic gas $800 \,cal$ heat energy is given at constant pressure. If vibrational mode is neglected, then energy used by gas in work done against surroundings is ......... $cal$
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A hydrogen cylinder is designed to withstand an internal pressure of $100 \,atm$. At $27^{\circ} C$, hydrogen is pumped into the cylinder which exerts a pressure of $20 \,atm$. At what temperature does the danger of explosion first sets in ......... $K$View Solution
- 2A vessel contains a mixture of one mole of oxygen and two moles of nitrogen at $300 K$. The ratio of the average rotational kinetic energy per ${O_2}$ molecule to that per ${N_2}$ molecule isView Solution
- 3The average translational kinetic energy of ${N}_{2}$ gas molecules at $\ldots \ldots \ldots . .{ }^{\circ} {C}$ becomes equal to the ${K} . {E}$. of an electron accelerated from rest through a potential difference of $0.1$ $volt.$View Solution
$\left(\right.$ Given $\left.{k}_{{B}}=1.38 \times 10^{-23} \, {J} / {K}\right)$
(Fill the nearest integer).
- 4View SolutionBoyle's law holds for an ideal gas during
- 5An ideal gas has molecules with $5\,$ degrees of freedom. The ratio of specific heats at constant pressure $(C_p)$ and at constant volume $(C_v)$ isView Solution
- 6View SolutionA gas is enclosed in a closed pot. On keeping this pot in a train moving with high speed, the temperature of the gas
- 7One mole of a monoatomic ideal gas is expanded by a process described by $p V^3=C$, where $C$ is a constant. The heat capacity of the gas during the process is given by ( $R$ is the gas constant)View Solution
- 8View SolutionThe relation between two specific heats of a gas is
- 9The value closest to the thermal velocity of a Helium atom at room temperature $(300\,K)$in $ms^{-1}$ is $[k_B\, = 1 .4\times10^{-23}\,J/K;\, m_{He}\, = 7\times10^{-27}\,kg]$View Solution
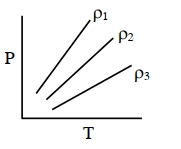
- 10$P-T$ diagram of an ideal gas having three different densities $\rho_1, \rho_2, \rho_3$ (in three different cases) is shown in the figure. Which of the following is correct :View Solution